Age and sex effects on paired-pulse suppression and prepulse inhibition of auditory evoked potentials
- PMID: 38655109
- PMCID: PMC11035799
- DOI: 10.3389/fnins.2024.1378619
Age and sex effects on paired-pulse suppression and prepulse inhibition of auditory evoked potentials
Abstract
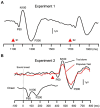
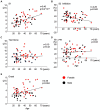
Responses to a sensory stimulus are inhibited by a preceding stimulus; if the two stimuli are identical, paired-pulse suppression (PPS) occurs; if the preceding stimulus is too weak to reliably elicit the target response, prepulse inhibition (PPI) occurs. PPS and PPI represent excitability changes in neural circuits induced by the first stimulus, but involve different mechanisms and are impaired in different diseases, e.g., impaired PPS in schizophrenia and Alzheimer's disease and impaired PPI in schizophrenia and movement disorders. Therefore, these measures provide information on several inhibitory mechanisms that may have roles in clinical conditions. In the present study, PPS and PPI of the auditory change-related cortical response were examined to establish normative data on healthy subjects (35 females and 32 males, aged 19-70 years). We also investigated the effects of age and sex on PPS and PPI to clarify whether these variables need to be considered as biases. The test response was elicited by an abrupt increase in sound pressure in a continuous sound and was recorded by electroencephalography. In the PPS experiment, the two change stimuli to elicit the cortical response were a 15-dB increase from the background of 65 dB separated by 600 ms. In the PPI experiment, the prepulse and test stimuli were 2- and 10-dB increases, respectively, with an interval of 50 ms. The results obtained showed that sex exerted similar effects on the two measures, with females having stronger test responses and weaker inhibition. On the other hand, age exerted different effects: aging correlated with stronger test responses and weaker inhibition in the PPS experiment, but had no effects in the PPI experiment. The present results suggest age and sex biases in addition to normative data on PPS and PPI of auditory change-related potentials. PPS and PPI, as well as other similar paradigms, such as P50 gating, may have different and common mechanisms. Collectively, they may provide insights into the pathophysiologies of diseases with impaired inhibitory function.
Keywords: P50 gating; aging; change-related potential; sensory gating; sensory suppression.
Copyright © 2024 Inui, Takeuchi, Borgil, Shingaki, Sugiyama, Taniguchi, Nishihara, Watanabe, Suzuki, Motomura and Kida.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Prepulse inhibition of change-related P50m no correlation with P50m gating.Springerplus. 2013 Nov 1;2:588. doi: 10.1186/2193-1801-2-588. eCollection 2013. Springerplus. 2013. PMID: 24255871 Free PMC article.
-
New paradigm for auditory paired pulse suppression.PLoS One. 2017 May 18;12(5):e0177747. doi: 10.1371/journal.pone.0177747. eCollection 2017. PLoS One. 2017. PMID: 28542290 Free PMC article.
-
Effects of acute nicotine on prepulse inhibition of auditory change-related cortical responses.Behav Brain Res. 2013 Nov 1;256:27-35. doi: 10.1016/j.bbr.2013.07.045. Epub 2013 Aug 6. Behav Brain Res. 2013. PMID: 23933145
-
Prepulse inhibition of the acoustic startle reflex and P50 gating in aging and alzheimer's disease.Ageing Res Rev. 2020 May;59:101028. doi: 10.1016/j.arr.2020.101028. Epub 2020 Feb 21. Ageing Res Rev. 2020. PMID: 32092463 Review.
-
Human studies of prepulse inhibition of startle: normal subjects, patient groups, and pharmacological studies.Psychopharmacology (Berl). 2001 Jul;156(2-3):234-58. doi: 10.1007/s002130100810. Psychopharmacology (Berl). 2001. PMID: 11549226 Review.
Cited by
-
Visual change-related brain potentials elicited by changes in doll hair color in school-aged children.BMC Neurosci. 2025 Aug 11;26(1):50. doi: 10.1186/s12868-025-00970-8. BMC Neurosci. 2025. PMID: 40790464 Free PMC article.
-
Short-latency prepulse inhibition of the trigeminal blink reflex.Front Neurosci. 2024 May 22;18:1357368. doi: 10.3389/fnins.2024.1357368. eCollection 2024. Front Neurosci. 2024. PMID: 38841093 Free PMC article.
References
-
- Adler L. E., Pachtman E., Franks R. D., Pecevich M., Waldo M. C., Freedman R. (1982). Neurophysiological evidence for a defect in neuronal mechanisms involved in sensory gating in schizophrenia. Biol. Psychiatry 17, 639–654. PMID: - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

