Automated Quantitative CD8+ Tumor-Infiltrating Lymphocytes and Tumor Mutation Burden as Independent Biomarkers in Melanoma Patients Receiving Front-Line Anti-PD-1 Immunotherapy
- PMID: 38655867
- PMCID: PMC11224974
- DOI: 10.1093/oncolo/oyae054
Automated Quantitative CD8+ Tumor-Infiltrating Lymphocytes and Tumor Mutation Burden as Independent Biomarkers in Melanoma Patients Receiving Front-Line Anti-PD-1 Immunotherapy
Abstract
Background: CD8+ tumor-infiltrating lymphocyte (TIL) predicts response to anti-PD-(L)1 therapy. However, there remains no standardized method to assess CD8+ TIL in melanoma, and developing a specific, cost-effective, reproducible, and clinically actionable biomarker to anti-PD-(L)1 remains elusive. We report on the development of automatic CD8+ TIL density quantification via whole slide image (WSI) analysis in advanced melanoma patients treated with front-line anti-PD-1 blockade, and correlation immunotherapy response.
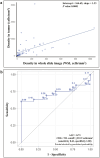
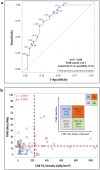
Methods: Seventy-eight patients treated with PD-1 inhibitors in the front-line setting between January 2015 and May 2023 at the University of Pittsburgh Cancer Institute were included. CD8+ TIL density was quantified using an image analysis algorithm on digitized WSI. Targeted next-generation sequencing (NGS) was performed to determine tumor mutation burden (TMB) in a subset of 62 patients. ROC curves were used to determine biomarker cutoffs and response to therapy. Correlation between CD8+ TIL density and TMB cutoffs and response to therapy was studied.
Results: Higher CD8+ TIL density was significantly associated with improved response to front-line anti-PD-1 across all time points measured. CD8+ TIL density ≥222.9 cells/mm2 reliably segregated responders and non-responders to front-line anti-PD-1 therapy regardless of when response was measured. In a multivariate analysis, patients with CD8+ TIL density exceeding cutoff had significantly improved PFS with a trend toward improved OS. Similarly, increasing TMB was associated with improved response to anti-PD-1, and a cutoff of 14.70 Mut/Mb was associated with improved odds of response. The correlation between TMB and CD8+ TIL density was low, suggesting that each represented independent predictive biomarkers of response.
Conclusions: An automatic digital analysis algorithm provides a standardized method to quantify CD8+ TIL density, which predicts response to front-line anti-PD-1 therapy. CD8+ TIL density and TMB are independent predictors of response to anti-PD-1 blockade.
Keywords: PD-1; PD-L1; TIL; TMB/Mb; biomarker; immunotherapy; melanoma; metastatic; tumor mutation burden.
© The Author(s) 2024. Published by Oxford University Press.
Conflict of interest statement
Hassane M. Zarour: grants/research support (institutional): NIH/NCI, Bristol-Myers Squibb and GlaxoSmithKline. Consultant: Checkmate Pharmaceuticals, GlaxoSmithKline, Bayer, and Vedanta. Intellectual Property: US Patent 63/124,231, “Compositions and Methods for Treating Cancer,” Dec 11, 2020. US Patent 63/208,719, “Compositions and Methods For Responsiveness to Immune Checkpoint Inhibitors (ICI), Increasing Effectiveness of ICI and Treating Cancer,” June 9, 2021. John M. Kirkwood: Grants/Research Support (institutional): Bristol-Myers Squibb, Amgen Inc. Consultant: Bristol-Myers Squibb, Checkmate Pharmaceuticals, Novartis, Amgen Inc., Checkmate, Castle Biosciences, Inc., Immunocore LLC, Iovance, Novartis. Diwakar Davar: Grants/Research Support (institutional): Arcus, CellSight Technologies, Immunocore, Merck, Regeneron Pharmaceuticals Inc., Tesaro/GSK. Consultant: ACM Bio, Ascendis Pharma, Clinical Care Options (CCO), Gerson Lehrman Group (GLG), Merck, Medical Learning Group (MLG), Xilio Therapeutics. CE Speakers’ Bureau: Castle Biosciences. Stockholder: None. Intellectual Property: US Patent 63/124,231, “Compositions and Methods for Treating Cancer,” Dec 11, 2020. US Patent 63/208,719, “Compositions and Methods For Responsiveness to Immune Checkpoint Inhibitors (ICI), Increasing Effectiveness of ICI and Treating Cancer,” June 9, 2021. The other authors indicated no financial relationships.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

