Postsynaptic potentials of soleus motor neurons produced by transspinal stimulation: a human single-motor unit study
- PMID: 38656134
- PMCID: PMC11381115
- DOI: 10.1152/jn.00077.2024
Postsynaptic potentials of soleus motor neurons produced by transspinal stimulation: a human single-motor unit study
Abstract
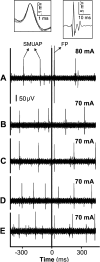
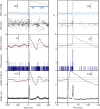
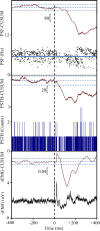
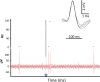
Transspinal (or transcutaneous spinal cord) stimulation is a noninvasive, cost-effective, easily applied method with great potential as a therapeutic modality for recovering somatic and nonsomatic functions in upper motor neuron disorders. However, how transspinal stimulation affects motor neuron depolarization is poorly understood, limiting the development of effective transspinal stimulation protocols for rehabilitation. In this study, we characterized the responses of soleus α motor neurons to single-pulse transspinal stimulation using single-motor unit (SMU) discharges as a proxy given the 1:1 discharge activation between the motor neuron and the motor unit. Peristimulus time histogram, peristimulus frequencygram, and surface electromyography (sEMG) were used to characterize the postsynaptic potentials of soleus motor neurons. Transspinal stimulation produced short-latency excitatory postsynaptic potentials (EPSPs) followed by two distinct phases of inhibitory postsynaptic potentials (IPSPs) in most soleus motor neurons and only IPSPs in others. Transspinal stimulation generated double discharges at short interspike intervals in a few motor units. The short-latency EPSPs were likely mediated by muscle spindle group Ia and II afferents, and the IPSPs via excitation of group Ib afferents and recurrent collaterals of motor neurons leading to activation of diverse spinal inhibitory interneuronal circuits. Further studies are warranted to understand better how transspinal stimulation affects depolarization of α motor neurons over multiple spinal segments. This knowledge will be seminal for developing effective transspinal stimulation protocols in upper motor neuron lesions.NEW & NOTEWORTHY Transspinal stimulation produces distinct actions on soleus motor neurons: an early short-latency excitation followed by two inhibitions or only inhibition and doublets. These results show how transspinal stimulation affects depolarization of soleus α motor neurons in healthy humans.
Keywords: motoneurons; motor units; postsynaptic potentials; transcutaneous spinal cord stimulation; transspinal stimulation.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures

References
-
- Gorodnichev RM, Pivovarova EA, Puhov A, Moiseev SA, Savochin AA, Moshonkina TR, Chsherbakova NA, Kilimnik VA, Selionov VA, Kozlovskaya IB, Edgerton VR, Gerasimenko YP. Transcutaneous electrical stimulation of the spinal cord: a noninvasive tool for the activation of stepping pattern generators in humans. Hum Physiol 38: 158–167, 2012. doi: 10.1134/S0362119712020065. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

