This is a preprint.
Glycogen phase separation drives macromolecular rearrangement and asymmetric division in E. coli
- PMID: 38659787
- PMCID: PMC11042326
- DOI: 10.1101/2024.04.19.590186
Glycogen phase separation drives macromolecular rearrangement and asymmetric division in E. coli
Abstract
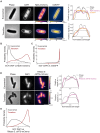
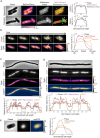
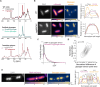
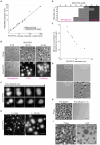
Bacteria often experience nutrient limitation in nature and the laboratory. While exponential and stationary growth phases are well characterized in the model bacterium Escherichia coli, little is known about what transpires inside individual cells during the transition between these two phases. Through quantitative cell imaging, we found that the position of nucleoids and cell division sites becomes increasingly asymmetric during transition phase. These asymmetries were coupled with spatial reorganization of proteins, ribosomes, and RNAs to nucleoid-centric localizations. Results from live-cell imaging experiments, complemented with genetic and 13C whole-cell nuclear magnetic resonance spectroscopy studies, show that preferential accumulation of the storage polymer glycogen at the old cell pole leads to the observed rearrangements and asymmetric divisions. In vitro experiments suggest that these phenotypes are likely due to the propensity of glycogen to phase separate in crowded environments, as glycogen condensates exclude fluorescent proteins under physiological crowding conditions. Glycogen-associated differences in cell sizes between strains and future daughter cells suggest that glycogen phase separation allows cells to store large glucose reserves without counting them as cytoplasmic space.
Keywords: Asymmetric division; bacteria; glycogen; nutrient limitation; phase separation.
Conflict of interest statement
DISCLOSURE AND COMPETING INTERESTS STATEMENT The authors declare no competing interests.
Figures

Similar articles
-
Biophysical Properties of Escherichia coli Cytoplasm in Stationary Phase by Superresolution Fluorescence Microscopy.mBio. 2020 Jun 16;11(3):e00143-20. doi: 10.1128/mBio.00143-20. mBio. 2020. PMID: 32546611 Free PMC article.
-
Genetic Manipulation of Glycogen Allocation Affects Replicative Lifespan in E. coli.PLoS Genet. 2016 Apr 19;12(4):e1005974. doi: 10.1371/journal.pgen.1005974. eCollection 2016 Apr. PLoS Genet. 2016. PMID: 27093302 Free PMC article.
-
Robustness of the Process of Nucleoid Exclusion of Protein Aggregates in Escherichia coli.J Bacteriol. 2016 Jan 4;198(6):898-906. doi: 10.1128/JB.00848-15. J Bacteriol. 2016. PMID: 26728194 Free PMC article.
-
The spatial biology of transcription and translation in rapidly growing Escherichia coli.Front Microbiol. 2015 Jul 2;6:636. doi: 10.3389/fmicb.2015.00636. eCollection 2015. Front Microbiol. 2015. PMID: 26191045 Free PMC article. Review.
-
Modulation of the nucleoid, the transcription apparatus, and the translation machinery in bacteria for stationary phase survival.Genes Cells. 1999 Mar;4(3):135-43. doi: 10.1046/j.1365-2443.1999.00247.x. Genes Cells. 1999. PMID: 10320479 Review.
References
-
- Alberti S & Hyman AA (2021) Biomolecular condensates at the nexus of cellular stress, protein aggregation disease and ageing. Nat Rev Mol Cell Biol 22: 196–213 - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
