AiZynth impact on medicinal chemistry practice at AstraZeneca
- PMID: 38665822
- PMCID: PMC11042116
- DOI: 10.1039/d3md00651d
AiZynth impact on medicinal chemistry practice at AstraZeneca
Abstract
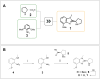
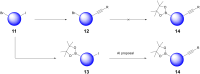
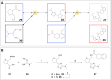
AstraZeneca chemists have been using the AI retrosynthesis tool AiZynth for three years. In this article, we present seven examples of how medicinal chemists using AiZynth positively impacted their drug discovery programmes. These programmes run the gamut from early-stage hit confirmation to late-stage route optimisation efforts. We also discuss the different use cases for which AI retrosynthesis tools are best suited.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
All authors of this article except YL and YZ are present or former employees of AstraZeneca and may have or have had a financial stake in the performance of the company.
Figures




References
-
- Jiang Y. Yu Y. Kong M. Mei Y. Yuan L. Huang Z. Kuang K. Wang Z. Yao H. Zou J. Coley C. W. Wei Y. Engineering. 2022;25:32–50. doi: 10.1016/j.eng.2022.04.021. - DOI
Publication types
LinkOut - more resources
Full Text Sources

