Characterization of PDGF-Induced Subcellular Calcium Regulation through Calcium Channels in Airway Smooth Muscle Cells by FRET Biosensors
- PMID: 38667172
- PMCID: PMC11048225
- DOI: 10.3390/bios14040179
Characterization of PDGF-Induced Subcellular Calcium Regulation through Calcium Channels in Airway Smooth Muscle Cells by FRET Biosensors
Abstract
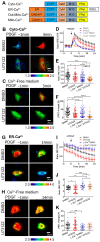
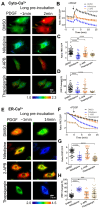
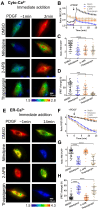
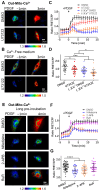
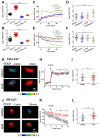
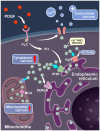
The homeostasis of cellular calcium is fundamental for many physiological processes, while the calcium levels remain inhomogeneous within cells. During the onset of asthma, epithelial and inflammatory cells secrete platelet-derived growth factor (PDGF), inducing the proliferation and migration of airway smooth muscle (ASM) to the epidermal layer, narrowing the airway. The regulation of ASM cells by PDGF is closely related to the conduction of calcium signals. In this work, we generated subcellular-targeted FRET biosensors to investigate calcium regulation in the different compartments of ASM cells. A PDGF-induced cytoplasmic calcium [Ca2+]C increase was attributed to both extracellular calcium influx and endoplasmic reticulum (ER) calcium [Ca2+]ER release, which was partially regulated by the PLC-IP3R pathway. Interestingly, the removal of the extracellular calcium influx led to inhibited ER calcium release, likely through inhibitory effects on the calcium-dependent activation of the ER ryanodine receptor. The inhibition of the L-type calcium channel on the plasma membrane or the SERCA pump on the ER resulted in both reduced [Ca2+]C and [Ca2+]ER from PDGF stimulation, while IP3R channel inhibition led to reduced [Ca2+]C only. The inhibited SERCA pump caused an immediate [Ca2+]C increase and [Ca2+]ER decrease, indicating active calcium exchange between the cytosol and ER storage in resting cells. PDGF-induced calcium at the outer mitochondrial membrane sub-region showed a similar regulatory response to cytosolic calcium, not influenced by the inhibition of the mitochondrial calcium uniporter channel. Therefore, our work identifies calcium flow pathways among the extracellular medium, cell cytosol, and ER via regulatory calcium channels. Specifically, extracellular calcium flow has an essential function in fully activating ER calcium release.
Keywords: FRET biosensor; airway smooth muscle cells; calcium channels; calcium signal; platelet-derived growth factor (PDGF); subcellular calcium regulation.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
STIM1 regulates platelet-derived growth factor-induced migration and Ca2+ influx in human airway smooth muscle cells.PLoS One. 2012;7(9):e45056. doi: 10.1371/journal.pone.0045056. Epub 2012 Sep 11. PLoS One. 2012. PMID: 22984609 Free PMC article.
-
Airway smooth muscle STIM1 and Orai1 are upregulated in asthmatic mice and mediate PDGF-activated SOCE, CRAC currents, proliferation, and migration.Pflugers Arch. 2012 Nov;464(5):481-92. doi: 10.1007/s00424-012-1160-5. Epub 2012 Sep 27. Pflugers Arch. 2012. PMID: 23014880 Free PMC article.
-
Differential estrogen-receptor activation regulates extracellular matrix deposition in human airway smooth muscle remodeling via NF-κB pathway.FASEB J. 2019 Dec;33(12):13935-13950. doi: 10.1096/fj.201901340R. Epub 2019 Oct 22. FASEB J. 2019. PMID: 31638834 Free PMC article.
-
Mitochondria Structure and Position in the Local Control of Calcium Signals in Smooth Muscle Cells.In: Trebak M, Earley S, editors. Signal Transduction and Smooth Muscle. Oxon (UK): CRC Press/Taylor & Francis; 2018 Aug 6. Chapter 9. In: Trebak M, Earley S, editors. Signal Transduction and Smooth Muscle. Oxon (UK): CRC Press/Taylor & Francis; 2018 Aug 6. Chapter 9. PMID: 31436944 Free Books & Documents. Review.
-
From Stores to Sinks: Structural Mechanisms of Cytosolic Calcium Regulation.Adv Exp Med Biol. 2017;981:215-251. doi: 10.1007/978-3-319-55858-5_10. Adv Exp Med Biol. 2017. PMID: 29594864 Review.
Cited by
-
FRET Visualization of High Mechanosensation of von Willebrand Factor to Hydrodynamic Force.Biosensors (Basel). 2025 Apr 14;15(4):248. doi: 10.3390/bios15040248. Biosensors (Basel). 2025. PMID: 40277561 Free PMC article.
References
-
- Bidaux G., Flourakis M., Thebault S., Zholos A., Beck B., Gkika D., Roudbaraki M., Bonnal J.L., Mauroy B., Shuba Y., et al. Prostate cell differentiation status determines transient receptor potential melastatin member 8 channel subcellular localization and function. J. Clin. Investig. 2007;117:1647–1657. doi: 10.1172/JCI30168. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

