Advancements and Challenges in Hydrogel Engineering for Regenerative Medicine
- PMID: 38667657
- PMCID: PMC11049258
- DOI: 10.3390/gels10040238
Advancements and Challenges in Hydrogel Engineering for Regenerative Medicine
Abstract
This manuscript covers the latest advancements and persisting challenges in the domain of tissue engineering, with a focus on the development and engineering of hydrogel scaffolds. It highlights the critical role of these scaffolds in emulating the native tissue environment, thereby providing a supportive matrix for cell growth, tissue integration, and reducing adverse reactions. Despite significant progress, this manuscript emphasizes the ongoing struggle to achieve an optimal balance between biocompatibility, biodegradability, and mechanical stability, crucial for clinical success. It also explores the integration of cutting-edge technologies like 3D bioprinting and biofabrication in constructing complex tissue structures, alongside innovative materials and techniques aimed at enhancing tissue growth and functionality. Through a detailed examination of these efforts, the manuscript sheds light on the potential of hydrogels in advancing regenerative medicine and the necessity for multidisciplinary collaboration to navigate the challenges ahead.
Keywords: biofabrication; hydrogel technology; regenerative medicine; stem cell differentiation; tissue engineering.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


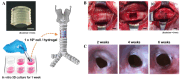
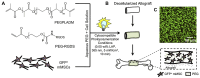
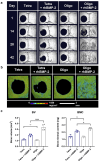
References
-
- Campos F., Bonhome-Espinosa A.B., Garcia-Martinez L., Duran J.D., Lopez-Lopez M.T., Alaminos M., Sanchez-Quevedo M.C., Carriel V. Ex vivo characterization of a novel tissue-like cross-linked fibrin-agarose hydrogel for tissue engineering applications. Biomed. Mater. 2016;11:055004. doi: 10.1088/1748-6041/11/5/055004. - DOI - PubMed
-
- Vedadghavami A., Minooei F., Mohammadi M.H., Khetani S., Rezaei Kolahchi A., Mashayekhan S., Sanati-Nezhad A. Manufacturing of hydrogel biomaterials with controlled mechanical properties for tissue engineering applications. Acta Biomater. 2017;62:42–63. doi: 10.1016/j.actbio.2017.07.028. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

