T Cell Receptor-Directed Bispecific T Cell Engager Targeting MHC-Linked NY-ESO-1 for Tumor Immunotherapy
- PMID: 38672132
- PMCID: PMC11048172
- DOI: 10.3390/biomedicines12040776
T Cell Receptor-Directed Bispecific T Cell Engager Targeting MHC-Linked NY-ESO-1 for Tumor Immunotherapy
Abstract
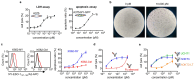
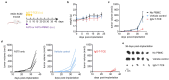
Antibody-based bispecific T cell engagers (TCEs) that redirect T cells to kill tumor cells have shown a promising therapeutic effect on hematologic malignancies. However, tumor-specific targeting is still a challenge for TCEs, impeding the development of TCEs for solid tumor therapy. The major histocompatibility complex (MHC) presents almost all intracellular peptides (including tumor-specific peptides) on the cell surface to be scanned by the TCR on T cells. With the premise of choosing optimal peptides, the final complex peptide-MHC could be the tumor-specific target for TCEs. Here, a novel TCR-directed format of a TCE targeting peptide-MHC was designed named IgG-T-TCE, which was modified from the IgG backbone and prepared in a mammalian cell expression system. The recombinant IgG-T-TCE-NY targeting NY-ESO-1157-165/HLA-A*02:01 could be generated in HEK293 cells with a glycosylated TCR and showed potency in T cell activation and redirecting T cells to specifically kill target tumor cells. We also found that the in vitro activity of IgG-T-TCE-NY could be leveraged by various anti-CD3 antibodies and Fc silencing. The IgG-T-TCE-NY efficiently inhibited tumor growth in a tumor-PBMC co-engrafted mouse model without any obvious toxicities.
Keywords: NY-ESO-1; T cell receptor; bispecific T cell engager; immunotherapy; pMHC; soluble expression.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
IL-2-armored peptide-major histocompatibility class I bispecific antibodies redirect antiviral effector memory CD8+ T cells to induce potent anti-cancer cytotoxic activity with limited cytokine release.MAbs. 2024 Jan-Dec;16(1):2395499. doi: 10.1080/19420862.2024.2395499. Epub 2024 Aug 28. MAbs. 2024. PMID: 39205483 Free PMC article.
-
Soluble Expression of Fc-Fused T Cell Receptors Allows Yielding Novel Bispecific T Cell Engagers.Biomedicines. 2021 Jul 8;9(7):790. doi: 10.3390/biomedicines9070790. Biomedicines. 2021. PMID: 34356854 Free PMC article.
-
T-cell-engaging antibodies for the treatment of solid tumors: challenges and opportunities.Curr Opin Oncol. 2022 Sep 1;34(5):552-558. doi: 10.1097/CCO.0000000000000869. Epub 2022 Jul 28. Curr Opin Oncol. 2022. PMID: 35880455 Free PMC article. Review.
-
ABBV-184: A Novel Survivin-specific TCR/CD3 Bispecific T-cell Engager is Active against Both Solid Tumor and Hematologic Malignancies.Mol Cancer Ther. 2023 Aug 1;22(8):903-912. doi: 10.1158/1535-7163.MCT-22-0770. Mol Cancer Ther. 2023. PMID: 37294945
-
T-Cell Engagers-The Structure and Functional Principle and Application in Hematological Malignancies.Cancers (Basel). 2024 Apr 20;16(8):1580. doi: 10.3390/cancers16081580. Cancers (Basel). 2024. PMID: 38672662 Free PMC article. Review.
Cited by
-
Recent Advancements in Cell-Based Therapies in Melanoma.Int J Mol Sci. 2024 Sep 12;25(18):9848. doi: 10.3390/ijms25189848. Int J Mol Sci. 2024. PMID: 39337333 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

