Deoxyxylulose 5-Phosphate Synthase Does Not Play a Major Role in Regulating the Methylerythritol 4-Phosphate Pathway in Poplar
- PMID: 38673766
- PMCID: PMC11049974
- DOI: 10.3390/ijms25084181
Deoxyxylulose 5-Phosphate Synthase Does Not Play a Major Role in Regulating the Methylerythritol 4-Phosphate Pathway in Poplar
Abstract
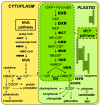
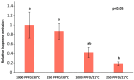
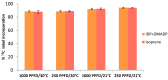
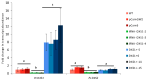
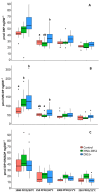
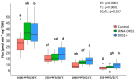
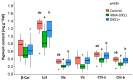
The plastidic 2-C-methylerythritol 4-phosphate (MEP) pathway supplies the precursors of a large variety of essential plant isoprenoids, but its regulation is still not well understood. Using metabolic control analysis (MCA), we examined the first enzyme of this pathway, 1-deoxyxylulose 5-phosphate synthase (DXS), in multiple grey poplar (Populus × canescens) lines modified in their DXS activity. Single leaves were dynamically labeled with 13CO2 in an illuminated, climate-controlled gas exchange cuvette coupled to a proton transfer reaction mass spectrometer, and the carbon flux through the MEP pathway was calculated. Carbon was rapidly assimilated into MEP pathway intermediates and labeled both the isoprene released and the IDP+DMADP pool by up to 90%. DXS activity was increased by 25% in lines overexpressing the DXS gene and reduced by 50% in RNA interference lines, while the carbon flux in the MEP pathway was 25-35% greater in overexpressing lines and unchanged in RNA interference lines. Isoprene emission was also not altered in these different genetic backgrounds. By correlating absolute flux to DXS activity under different conditions of light and temperature, the flux control coefficient was found to be low. Among isoprenoid end products, isoprene itself was unchanged in DXS transgenic lines, but the levels of the chlorophylls and most carotenoids measured were 20-30% less in RNA interference lines than in overexpression lines. Our data thus demonstrate that DXS in the isoprene-emitting grey poplar plays only a minor part in controlling flux through the MEP pathway.
Keywords: DMADP; DXS enzyme; IDP; MEP pathway; flux control coefficient (FCC); isoprene; isoprenoid; metabolic control analysis (MCA).
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Analysis of 1-deoxy-D-xylulose 5-phosphate synthase activity in Grey poplar leaves using isotope ratio mass spectrometry.Phytochemistry. 2010 Jun;71(8-9):918-22. doi: 10.1016/j.phytochem.2010.02.016. Epub 2010 Mar 18. Phytochemistry. 2010. PMID: 20303132
-
Metabolic flux analysis of plastidic isoprenoid biosynthesis in poplar leaves emitting and nonemitting isoprene.Plant Physiol. 2014 May;165(1):37-51. doi: 10.1104/pp.114.236018. Epub 2014 Mar 3. Plant Physiol. 2014. PMID: 24590857 Free PMC article.
-
Feedback inhibition of deoxy-D-xylulose-5-phosphate synthase regulates the methylerythritol 4-phosphate pathway.J Biol Chem. 2013 Jun 7;288(23):16926-16936. doi: 10.1074/jbc.M113.464636. Epub 2013 Apr 23. J Biol Chem. 2013. PMID: 23612965 Free PMC article.
-
Methylerythritol 4-phosphate (MEP) pathway metabolic regulation.Nat Prod Rep. 2014 Aug;31(8):1043-55. doi: 10.1039/c3np70124g. Nat Prod Rep. 2014. PMID: 24921065 Review.
-
An account of cloned genes of Methyl-erythritol-4-phosphate pathway of isoprenoid biosynthesis in plants.Curr Issues Mol Biol. 2009;11 Suppl 1:i35-45. Epub 2009 Feb 2. Curr Issues Mol Biol. 2009. PMID: 19193963 Review.
References
-
- Croteau R., Kutchan T.M., Lewis N.G. Natural Products (Secondary Metabolites) Biochem. Mol. Biol. Plants. 2000;24:1250–1319.
-
- Croteau R. Biosynthesis and Catabolism of Monoterpenoids. Chem. Rev. 1987;87:929–954. doi: 10.1021/cr00081a004. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

