Scandium Ion-Promoted Electron-Transfer Disproportionation of 2-Phenyl-4,4,5,5-tetramethylimidazoline-1-oxyl 3-Oxide (PTIO•) in Acetonitrile and Its Regeneration Induced by Water
- PMID: 38674002
- PMCID: PMC11050215
- DOI: 10.3390/ijms25084417
Scandium Ion-Promoted Electron-Transfer Disproportionation of 2-Phenyl-4,4,5,5-tetramethylimidazoline-1-oxyl 3-Oxide (PTIO•) in Acetonitrile and Its Regeneration Induced by Water
Abstract
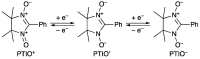
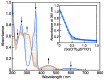
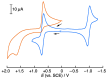
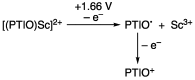
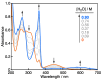
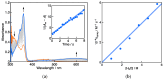
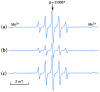
2-Phenyl-4,4,5,5-tetramethylimidazoline-1-oxyl 3-oxide (PTIO•), a persistent nitronyl nitroxide radical, has been used for the detection and trapping of nitric oxide, as a redox mediator for batteries, for the activity estimation of antioxidants, and so on. However, there is no report on the reactivity of PTIO• in the presence of redox-inactive metal ions. In this study, it is demonstrated that the addition of scandium triflate, Sc(OTf)3 (OTf = OSO2CF3), to an acetonitrile (MeCN) solution of PTIO• resulted in an electron-transfer disproportionation to generate the corresponding cation (PTIO+) and anion (PTIO-), the latter of which is suggested to be stabilized by Sc3+ to form [(PTIO)Sc]2+. The decay of the absorption band at 361 nm due to PTIO•, monitored using a stopped-flow technique, obeyed second-order kinetics. The second-order rate constant for the disproportionation, thus determined, increased with increasing the Sc(OTf)3 concentration to reach a constant value. A drastic change in the cyclic voltammogram recorded for PTIO• in deaerated MeCN containing 0.10 M Bu4NClO4 was also observed upon addition of Sc(OTf)3, suggesting that the large positive shift of the one-electron reduction potential of PTIO• (equivalent to the one-electron oxidation potential of PTIO-) in the presence of Sc(OTf)3 may result in the disproportionation. When H2O was added to the PTIO•-Sc(OTf)3 system in deaerated MeCN, PTIO• was completely regenerated. It is suggested that the complex formation of Sc3+ with H2O may weaken the interaction between PTIO- and Sc3+, leading to electron-transfer comproportionation to regenerate PTIO•. The reversible disproportionation of PTIO• was also confirmed by electron paramagnetic resonance (EPR) spectroscopy.
Keywords: Lewis acid; comproportionation; cyclic voltammetry; disproportionation; electron paramagnetic resonance; electron transfer; kinetics; radical; reaction mechanism; scandium ion.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
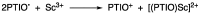

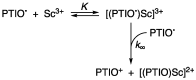
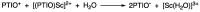
Similar articles
-
Water-Induced Regeneration of a 2,2-Diphenyl-1-picrylhydrazyl Radical after Its Scandium Ion-Promoted Electron-Transfer Disproportionation in an Aprotic Medium.Molecules. 2023 Jun 26;28(13):5002. doi: 10.3390/molecules28135002. Molecules. 2023. PMID: 37446663 Free PMC article.
-
On the distinction between nitroxyl and nitric oxide using nitronyl nitroxides.J Am Chem Soc. 2010 Jun 23;132(24):8428-32. doi: 10.1021/ja101945j. J Am Chem Soc. 2010. PMID: 20504018
-
Metal ion-catalyzed cycloaddition vs hydride transfer reactions of NADH analogues with p-benzoquinones.J Am Chem Soc. 2001 Oct 24;123(42):10191-9. doi: 10.1021/ja016370k. J Am Chem Soc. 2001. PMID: 11603968
-
Scandium ion-promoted photoinduced electron transfer from electron donors to acridine and pyrene. Essential role of scandium ion in photocatalytic oxygenation of hexamethylbenzene.J Am Chem Soc. 2004 Jun 23;126(24):7585-94. doi: 10.1021/ja031649h. J Am Chem Soc. 2004. PMID: 15198606
-
Scandium ion-promoted reduction of heterocyclic N=N double bond. Hydride transfer vs electron transfer.J Am Chem Soc. 2002 Oct 23;124(42):12566-73. doi: 10.1021/ja026592y. J Am Chem Soc. 2002. PMID: 12381201
References
-
- Fukuzumi S., Ohkubo K. Metal ion-coupled and decoupled electron transfer. Coord. Chem. Rev. 2010;254:372–385. doi: 10.1016/j.ccr.2009.10.020. - DOI
-
- Ghosh T.K., Maity S., Ghosh S., Gomila R.M., Frontera A., Ghosh A. Role of redox-inactive metal ions in modulating the reduction potential of uranyl Schiff base complexes: Detailed experimental and theoretical studies. Inorg. Chem. 2022;61:7130–7142. doi: 10.1021/acs.inorgchem.2c00645. - DOI - PubMed
-
- Mukai K., Nakamura A., Nagaoka S., Ouchi A., Azuma N. Notable effects of the metal salts on the quenching reaction of singlet oxygen by α-tocopherol in ethanol solution. Bull. Chem. Soc. Jpn. 2015;88:1503–1510. doi: 10.1246/bcsj.20150182. - DOI
-
- Mukai K., Kohno Y., Ouchi A., Nagaoka S. Notable effect of metal salts on UV-vis absorption spectra of α-, β-, γ-, and δ-tocopheroxyl radicals in acetonitrile solution. The complex formation between tocopheroxyls and metal cations. J. Phys. Chem. B. 2012;116:8930–8941. doi: 10.1021/jp305039z. - DOI - PubMed
-
- Mukai K., Oi M., Ouchi A., Nagaoka S. Kinetic study of the α-tocopherol-regeneration reaction of ubiquinol-10 in methanol and acetonitrile solutions: Notable effect of the alkali and alkaline earth metal salts on the reaction rates. J. Phys. Chem. B. 2012;116:2615–2621. doi: 10.1021/jp2118086. - DOI - PubMed
MeSH terms
Substances
Grants and funding
- JP18K06620/Ministry of Education, Culture, Sports, Science and Technology
- JP20H02779/Ministry of Education, Culture, Sports, Science and Technology
- JP20H04819/Ministry of Education, Culture, Sports, Science and Technology
- JP18H04650/Ministry of Education, Culture, Sports, Science and Technology
- JP17H03010/Ministry of Education, Culture, Sports, Science and Technology
LinkOut - more resources
Full Text Sources

