Reservoir displacement by an invasive rodent reduces Lassa virus zoonotic spillover risk
- PMID: 38678025
- PMCID: PMC11055883
- DOI: 10.1038/s41467-024-47991-1
Reservoir displacement by an invasive rodent reduces Lassa virus zoonotic spillover risk
Abstract
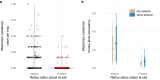
The black rat (Rattus rattus) is a globally invasive species that has been widely introduced across Africa. Within its invasive range in West Africa, R. rattus may compete with the native rodent Mastomys natalensis, the primary reservoir host of Lassa virus, a zoonotic pathogen that kills thousands annually. Here, we use rodent trapping data from Sierra Leone and Guinea to show that R. rattus presence reduces M. natalensis density within the human dwellings where Lassa virus exposure is most likely to occur. Further, we integrate infection data from M. natalensis to demonstrate that Lassa virus zoonotic spillover risk is lower at sites with R. rattus. While non-native species can have numerous negative effects on ecosystems, our results suggest that R. rattus invasion has the indirect benefit of decreasing zoonotic spillover of an endemic pathogen, with important implications for invasive species control across West Africa.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

