Semaphorin 7A promotes endothelial permeability and inflammation via plexin C1 and integrin β1 in Kawasaki disease
- PMID: 38678170
- PMCID: PMC11055240
- DOI: 10.1186/s12887-024-04766-3
Semaphorin 7A promotes endothelial permeability and inflammation via plexin C1 and integrin β1 in Kawasaki disease
Abstract
Background: Kawasaki disease (KD) is a pediatric systemic vasculitis characterized by endothelial cell dysfunction. Semaphorin 7A (Sema7A) has been reported to regulate endothelial phenotypes associated with cardiovascular diseases, while its role in KD remains unknown. This study aims to investigate the effect of Sema7A on endothelial permeability and inflammatory response in KD conditions.
Methods: Blood samples were collected from 68 KD patients and 25 healthy children (HC). The levels of Sema7A and A Disintegrin and Metalloprotease 17 (ADAM17) in serum were measured by enzyme-linked immunosorbent assay (ELISA), and Sema7A expression in blood cells was analyzed by flow cytometry. Ex vivo monocytes were used for Sema7A shedding assays. In vitro human coronary artery endothelial cells (HCAECs) were cultured in KD sera and stimulated with Sema7A, and TNF-α, IL-1β, IL-6, and IL-18 of HCAECs were measured by ELISA and qRT-PCR. HCAECs monolayer permeability was measured by FITC-dextran.
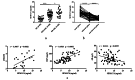
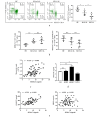
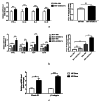
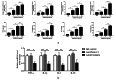
Results: The serum level of Sema7A was significantly higher in KD patients than in HC and correlated with disease severity. Monocytes were identified as one of the source of elevated serum Sema7A, which implicates a process of ADAM17-dependent shedding. Sera from KD patients induced upregulation of plexin C1 and integrin β1 in HCAECs compared to sera from HC. Sema7A mediated the proinflammatory cytokine production of HCAECs in an integrin β1-dependent manner, while both plexin C1 and integrin β1 contributed to Sema7A-induced HCAEC hyperpermeability.
Conclusions: Sema7A is involved in the progression of KD vasculitis by promoting endothelial permeability and inflammation through a plexin C1 and integrin β1-dependent pathway. Sema7A may serve as a potential biomarker and therapeutic target in the prognosis and treatment of KD.
Keywords: Endothelial cell; Inflammation; Kawasaki disease; Semaphorin 7A.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Neutrophil-Derived Semaphorin 4D Induces Inflammatory Cytokine Production of Endothelial Cells via Different Plexin Receptors in Kawasaki Disease.Biomed Res Int. 2020 Dec 16;2020:6663291. doi: 10.1155/2020/6663291. eCollection 2020. Biomed Res Int. 2020. PMID: 33381571 Free PMC article.
-
Endothelial semaphorin 7A promotes seawater aspiration-induced acute lung injury through plexin C1 and β1 integrin.Mol Med Rep. 2017 Oct;16(4):4215-4221. doi: 10.3892/mmr.2017.7097. Epub 2017 Jul 27. Mol Med Rep. 2017. PMID: 28765893
-
Vascular Semaphorin 7A Upregulation by Disturbed Flow Promotes Atherosclerosis Through Endothelial β1 Integrin.Arterioscler Thromb Vasc Biol. 2018 Feb;38(2):335-343. doi: 10.1161/ATVBAHA.117.310491. Epub 2017 Dec 21. Arterioscler Thromb Vasc Biol. 2018. PMID: 29269512 Free PMC article.
-
SEMA7A: A glycoprotein with therapeutic potential in inflammatory diseases and tumor development.Int J Biol Macromol. 2025 Jul;318(Pt 2):144907. doi: 10.1016/j.ijbiomac.2025.144907. Epub 2025 Jun 4. Int J Biol Macromol. 2025. PMID: 40480556 Review.
-
The involvement of semaphorin 7A in tumorigenic and immunoinflammatory regulation.J Cell Physiol. 2021 Sep;236(9):6235-6248. doi: 10.1002/jcp.30340. Epub 2021 Feb 21. J Cell Physiol. 2021. PMID: 33611799 Review.
Cited by
-
Immunophenotype of Kawasaki Disease: Insights into Pathogenesis and Treatment Response.Life (Basel). 2025 Jun 25;15(7):1012. doi: 10.3390/life15071012. Life (Basel). 2025. PMID: 40724515 Free PMC article. Review.
-
NK/DC crosstalk-modulating antitumor activity via Sema3E/PlexinD1 axis for enhanced cancer immunotherapy.Immunol Res. 2024 Dec;72(6):1217-1228. doi: 10.1007/s12026-024-09536-y. Epub 2024 Sep 5. Immunol Res. 2024. PMID: 39235526 Review.
-
Up-regulated vitronectin in Kawasaki disease shock syndrome serves as a potential biomarker.Transl Pediatr. 2025 Jun 27;14(6):1230-1244. doi: 10.21037/tp-2025-159. Epub 2025 Jun 25. Transl Pediatr. 2025. PMID: 40688201 Free PMC article.
-
Semaphorin 7a is protective through immune modulation during acetaminophen-induced liver injury.J Inflamm (Lond). 2025 Mar 20;22(1):13. doi: 10.1186/s12950-025-00429-x. J Inflamm (Lond). 2025. PMID: 40114253 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
- 2023-JC-YB-718/Natural Science Foundation of Shaanxi Province
- 2024JC-YBQN-0869/Natural Science Foundation of Shaanxi Province
- 202222/Science and Technology Plan Project of Weiyang District Xi'an City
- 2022NLTS107/Scientific Research Capacity Improvement Project of Xi'an Medical University
- 2023qn12/Scientific Research Project of Xi'an Health Commission
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

