Reasons, Efficacy and Safety of Switching to Dolutegravir-Based Regimens Among Virologically Suppressed PLWH: A Retrospective Cohort Study of 96 Weeks
- PMID: 38681898
- PMCID: PMC11055531
- DOI: 10.2147/IDR.S451346
Reasons, Efficacy and Safety of Switching to Dolutegravir-Based Regimens Among Virologically Suppressed PLWH: A Retrospective Cohort Study of 96 Weeks
Abstract
Purpose: The study aimed to explore the reasons, efficacy, and safety of switching to dolutegravir (DTG) based regimens in virologically suppressed people living with HIV (PLWH) in tertiary hospitals in China. Therefore, the study could provide a valuable reference for the rational clinical use of DTG.
Methods: PLWH's basic information, treatment details, and reasons for switching were collected, through the electrical clinical medical record system and telephone follow-up. Data included the proportion of PLWH with HIV RNA <50 copies/mL, changes in immunological indicators, and metabolic metrics at week 48 and week 96.
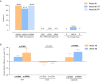
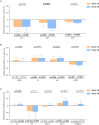
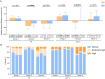
Results: 319 PLWH were included in the analysis. The three major reasons for switching were neurological toxicity (16.30%), simplification (13.79%), and renal toxicity (11.29%). Our study showed high rates of virologic suppression in the per-protocol analysis (week 48: 99.69%; week 96: 99.29%) after switching to DTG-based regimens. The median CD4+ T cell count increased from 579 cells/μL (IQR 420.5-758) to 642 cells/μL (IQR 466.5-854) at week 96 (p<0.0001). An improvement was observed in liver function (ALT: p<0.0001; AST: p<0.0001) and fasting glucose (p<0.0001). However, there was an elevation in creatinine (Cr) (p<0.0001) and a slight decrease in the estimated glomerular filtration rate (eGFR) (p<0.0001). Regarding lipid profile, triglyceride (TG) levels declined, while total cholesterol (TC) and low-density lipoprotein cholesterol (LDL-C) levels increased. Further analysis revealed that the increase in TC and LDL-C was associated with the withdrawal of tenofovir disoproxil fumarate (TDF). This observed increase in lipid parameters only concerned the PLWH who switched from a TDF-containing regimen to a non-TDF regimen.
Conclusion: This study confirmed the virologic efficacy of switching to DTG-based regimens in virologically suppressed PLWH over a 96-week period. The findings also expanded the evidence of immune reconstitution and metabolic safety associated with this switch.
Keywords: HIV; dolutegravir; lipids profile; switching; virologically suppressed.
© 2024 Deng et al.
Conflict of interest statement
The authors declare no conflicts of interest in this work.
Figures


References
-
- World Health Organization. WHO recommends dolutegravir as preferred HIV treatment option in all populations. Available from: https://www.who.int/news/item/22-07-2019-whorecommends-dolutegravir-as-p.... Accessed September 23, 2022.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

