Phenylephrine alters phase synchronization between cerebral blood velocity and blood pressure in ME/CFS with orthostatic intolerance
- PMID: 38682242
- PMCID: PMC11381003
- DOI: 10.1152/ajpregu.00071.2024
Phenylephrine alters phase synchronization between cerebral blood velocity and blood pressure in ME/CFS with orthostatic intolerance
Abstract
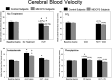
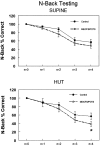
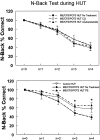
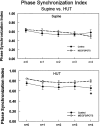
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) with orthostatic intolerance (OI) is characterized by neurocognitive deficits perhaps related to upright hypocapnia and loss of cerebral autoregulation (CA). We performed N-back neurocognition testing and calculated the phase synchronization index (PhSI) between arterial pressure (AP) and cerebral blood velocity (CBV) as a time-dependent measurement of cerebral autoregulation in 11 control (mean age = 24.1 yr) and 15 patients with ME/CFS (mean age = 21.8 yr). All patients with ME/CFS had postural tachycardia syndrome (POTS). A 10-min 60° head-up tilt (HUT) significantly increased heart rate (109.4 ± 3.9 vs. 77.2 ± 1.6 beats/min, P < 0.05) and respiratory rate (20.9 ± 1.7 vs. 14.2 ± 1.2 breaths/min, P < 0.05) and decreased end-tidal CO2 (ETCO2; 33.9 ± 1.1 vs. 42.8 ± 1.2 Torr, P < 0.05) in ME/CFS versus control. In ME/CFS, HUT significantly decreased CBV compared with control (-22.5% vs. -8.7%, P < 0.005). To mitigate the orthostatic CBV reduction, we administered supplemental CO2, phenylephrine, and acetazolamide and performed N-back testing supine and during HUT. Only phenylephrine corrected the orthostatic decrease in neurocognition by reverting % correct n = 4 N-back during HUT in ME/CFS similar to control (ME/CFS = 38.5 ± 5.5 vs. ME/CFS + PE= 65.6 ± 5.7 vs. Control 56.9 ± 7.5). HUT in ME/CFS resulted in increased PhSI values indicating decreased CA. Although CO2 and acetazolamide had no effect on PhSI in ME/CFS, phenylephrine caused a significant reduction in PhSI (ME/CFS = 0.80 ± 0.03 vs. ME/CFS + PE= 0.69 ± 0.04, P < 0.05) and improved cerebral autoregulation. Thus, PE improved neurocognitive function in patients with ME/CFS, perhaps related to improved neurovascular coupling, cerebral autoregulation, and maintenance of CBV.NEW & NOTEWORTHY We evaluated cognitive function before and after CO2, acetazolamide, and phenylephrine, which mitigate orthostatic reductions in cerebral blood velocity. Neither CO2 nor acetazolamide affected N-back testing (% correct answers) during an orthostatic challenge. Only phenylephrine improved upright N-back performance in ME/CFS, as it both blocked hyperventilation and increased CO2 significantly compared with those untreated. And only phenylephrine resulted in improved PSI values in both ME/CFS and control while upright, suggesting improved cerebral autoregulation.
Keywords: N-back testing; myalgic encephalomyelitis/chronic fatigue syndrome; phase synchronization; phenylephrine.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures


Similar articles
-
Phenylephrine alteration of cerebral blood flow during orthostasis: effect on n-back performance in chronic fatigue syndrome.J Appl Physiol (1985). 2014 Nov 15;117(10):1157-64. doi: 10.1152/japplphysiol.00527.2014. Epub 2014 Oct 2. J Appl Physiol (1985). 2014. PMID: 25277740 Free PMC article. Clinical Trial.
-
Cerebral blood flow remains reduced after tilt testing in myalgic encephalomyelitis/chronic fatigue syndrome patients.Clin Neurophysiol Pract. 2021 Sep 23;6:245-255. doi: 10.1016/j.cnp.2021.09.001. eCollection 2021. Clin Neurophysiol Pract. 2021. PMID: 34667909 Free PMC article.
-
Postural neurocognitive and neuronal activated cerebral blood flow deficits in young chronic fatigue syndrome patients with postural tachycardia syndrome.Am J Physiol Heart Circ Physiol. 2012 Mar 1;302(5):H1185-94. doi: 10.1152/ajpheart.00994.2011. Epub 2011 Dec 16. Am J Physiol Heart Circ Physiol. 2012. PMID: 22180650 Free PMC article.
-
Cerebral Blood Flow in Orthostatic Intolerance.J Am Heart Assoc. 2025 Feb 4;14(3):e036752. doi: 10.1161/JAHA.124.036752. Epub 2025 Feb 3. J Am Heart Assoc. 2025. PMID: 39895557 Free PMC article. Review.
-
Tetrahydrobiopterin in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A Friend or Foe?Biomolecules. 2025 Jan 10;15(1):102. doi: 10.3390/biom15010102. Biomolecules. 2025. PMID: 39858496 Free PMC article. Review.
References
-
- Fukuda K, Straus SE, Hickie I, Sharpe MC, Komaroff A, Schluederberg A, Jones JF, Lloyd AR, Wessely S, Gantz NG, Holmes GP, Steele L, Reyes M, Abbey S, Rest J, Jolson H, Peterson DL, Vercoulen JHMM, Tirelli U, Evengard B, Natelson BH, Reeves WC. The chronic fatigue syndrome: a comprehensive approach to its definition and study. Ann Intern Med 121: 953–959, 1994. doi:10.7326/0003-4819-121-12-199412150-00009. - DOI - PubMed
-
- Committee on the Diagnostic Criteria for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome, Board on the Health of Select Populations, Institute of Medicine. Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness (Online). Washington, DC: National Academies Press (US). http://www.ncbi.nlm.nih.gov/books/NBK274235/ [2024 Apr 12].
-
- Stewart JM, Boris JR, Chelimsky G, Fischer PR, Fortunato JE, Grubb BP, Heyer GL, Jarjour IT, Medow MS, Numan MT, Pianosi PT, Singer W, Tarbell S, Chelimsky TC; Pediatric Writing Group of the American Autonomic Society. Pediatric disorders of orthostatic intolerance. Pediatrics 141: e20171673, 2018. doi:10.1542/peds.2017-1673. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

