Membrane lipid remodeling eradicates Helicobacter pylori by manipulating the cholesteryl 6'-acylglucoside biosynthesis
- PMID: 38685037
- PMCID: PMC11057186
- DOI: 10.1186/s12929-024-01031-8
Membrane lipid remodeling eradicates Helicobacter pylori by manipulating the cholesteryl 6'-acylglucoside biosynthesis
Abstract
Background: Helicobacter pylori, the main cause of various gastric diseases, infects approximately half of the human population. This pathogen is auxotrophic for cholesterol which it converts to various cholesteryl α-glucoside derivatives, including cholesteryl 6'-acyl α-glucoside (CAG). Since the related biosynthetic enzymes can be translocated to the host cells, the acyl chain of CAG likely comes from its precursor phosphatidylethanolamine (PE) in the host membranes. This work aims at examining how the acyl chain of CAG and PE inhibits the membrane functions, especially bacterial adhesion.
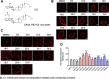
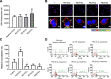
Methods: Eleven CAGs that differ in acyl chains were used to study the membrane properties of human gastric adenocarcinoma cells (AGS cells), including lipid rafts clustering (monitored by immunofluorescence with confocal microscopy) and lateral membrane fluidity (by the fluorescence recovery after photobleaching). Cell-based and mouse models were employed to study the degree of bacterial adhesion, the analyses of which were conducted by using flow cytometry and immunofluorescence staining, respectively. The lipidomes of H. pylori, AGS cells and H. pylori-AGS co-cultures were analyzed by Ultraperformance Liquid Chromatography-Tandem Mass Spectroscopy (UPLC-MS/MS) to examine the effect of PE(10:0)2, PE(18:0)2, PE(18:3)2, or PE(22:6)2 treatments.
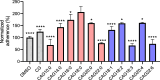
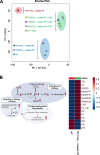
Results: CAG10:0, CAG18:3 and CAG22:6 were found to cause the most adverse effect on the bacterial adhesion. Further LC-MS analysis indicated that the treatment of PE(10:0)2 resulted in dual effects to inhibit the bacterial adhesion, including the generation of CAG10:0 and significant changes in the membrane compositions. The initial (1 h) lipidome changes involved in the incorporation of 10:0 acyl chains into dihydro- and phytosphingosine derivatives and ceramides. In contrast, after 16 h, glycerophospholipids displayed obvious increase in their very long chain fatty acids, monounsaturated and polyunsaturated fatty acids that are considered to enhance membrane fluidity.
Conclusions: The PE(10:0)2 treatment significantly reduced bacterial adhesion in both AGS cells and mouse models. Our approach of membrane remodeling has thus shown great promise as a new anti-H. pylori therapy.
Keywords: Helicobacter pylori; Adhesion; Biosynthesis; Cholesterol; Membrane; Phospholipids.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Enhanced enzymatic production of cholesteryl 6'-acylglucoside impairs lysosomal degradation for the intracellular survival of Helicobacter pylori.J Biomed Sci. 2021 Oct 27;28(1):72. doi: 10.1186/s12929-021-00768-w. J Biomed Sci. 2021. PMID: 34706729 Free PMC article.
-
Helicobacter pylori cholesteryl glucosides interfere with host membrane phase and affect type IV secretion system function during infection in AGS cells.Mol Microbiol. 2012 Jan;83(1):67-84. doi: 10.1111/j.1365-2958.2011.07910.x. Epub 2011 Nov 22. Mol Microbiol. 2012. PMID: 22053852
-
Metabolic labelling of cholesteryl glucosides in Helicobacter pylori reveals how the uptake of human lipids enhances bacterial virulence.Chem Sci. 2016 Sep 1;7(9):6208-6216. doi: 10.1039/c6sc00889e. Epub 2016 Jun 3. Chem Sci. 2016. PMID: 30034762 Free PMC article.
-
Therapeutic potential of cholesteryl O-acyl α-glucoside found in Helicobacter pylori.Curr Med Chem. 2012;19(28):4869-74. doi: 10.2174/092986712803341502. Curr Med Chem. 2012. PMID: 22934809 Review.
-
Unique responses of Helicobacter pylori to exogenous hydrophobic compounds.Chem Phys Lipids. 2020 Jul;229:104908. doi: 10.1016/j.chemphyslip.2020.104908. Epub 2020 Apr 4. Chem Phys Lipids. 2020. PMID: 32259519 Review.
Cited by
-
Challenges and Prospects for Eradication of Helicobacter pylori: Targeting Virulence Factors, Metabolism, and Vaccine Innovation.Pathogens. 2025 Jun 21;14(7):619. doi: 10.3390/pathogens14070619. Pathogens. 2025. PMID: 40732667 Free PMC article. Review.
-
The significance of calcium ions in cerebral ischemia-reperfusion injury: mechanisms and intervention strategies.Front Mol Biosci. 2025 May 12;12:1585758. doi: 10.3389/fmolb.2025.1585758. eCollection 2025. Front Mol Biosci. 2025. PMID: 40421420 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

