Single cell sequencing data identify distinct B cell and fibroblast populations in stricturing Crohn's disease
- PMID: 38685679
- PMCID: PMC11058334
- DOI: 10.1111/jcmm.18344
Single cell sequencing data identify distinct B cell and fibroblast populations in stricturing Crohn's disease
Abstract
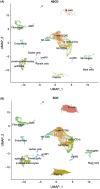
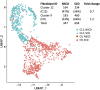
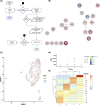
Single cell RNA sequencing of human full thickness Crohn's disease (CD) small bowel resection specimens was used to identify potential therapeutic targets for stricturing (S) CD. Using an unbiased approach, 16 cell lineages were assigned within 14,539 sequenced cells from patient-matched SCD and non-stricturing (NSCD) preparations. SCD and NSCD contained identical cell types. Amongst immune cells, B cells and plasma cells were selectively increased in SCD samples. B cell subsets suggested formation of tertiary lymphoid tissue in SCD and compared with NSCD there was an increase in IgG, and a decrease in IgA plasma cells, consistent with their potential role in CD fibrosis. Two Lumican-positive fibroblast subtypes were identified and subclassified based on expression of selectively enriched genes as fibroblast clusters (C) 12 and C9. Cells within these clusters expressed the profibrotic genes Decorin (C12) and JUN (C9). C9 cells expressed ACTA2; ECM genes COL4A1, COL4A2, COL15A1, COL6A3, COL18A1 and ADAMDEC1; LAMB1 and GREM1. GO and KEGG Biological terms showed extracellular matrix and stricture organization associated with C12 and C9, and regulation of WNT pathway genes with C9. Trajectory and differential gene analysis of C12 and C9 identified four sub-clusters. Intra sub-cluster gene analysis detected 13 co-regulated gene modules that aligned along predicted pseudotime trajectories. CXCL14 and ADAMDEC1 were key markers in module 1. Our findings support further investigation of fibroblast heterogeneity and interactions with local and circulating immune cells at earlier time points in fibrosis progression. Breaking these interactions by targeting one or other population may improve therapeutic management for SCD.
Keywords: B cells; Crohn's disease; fibroblasts; fibrosis; stricturing.
© 2024 The Authors. Journal of Cellular and Molecular Medicine published by Foundation for Cellular and Molecular Medicine and John Wiley & Sons Ltd.
Conflict of interest statement
The authors confirm there are no conflicts of interest.
Figures


References
-
- Van Linthout S, Miteva K, Tschöpe C. Crosstalk between fibroblasts and inflammatory cells. Cardiovasc Res. 2014;102:258‐269. - PubMed
-
- Chan WPW, Mourad F, Leong RW. Crohn's disease associated strictures. J Gastroenterol Hepatol. 2018;33:998‐1008. - PubMed
-
- Feagan BG, Bala M, Yan S, Olson A, Hanauer S. Unemployment and disability in patients with moderately to severely active Crohn's disease. J Clin Gastroenterol. 2005;39:390‐395. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

