Pre-soil fumigation with ammonium bicarbonate and lime modulates the rhizosphere microbiome to mitigate clubroot disease in Chinese cabbage
- PMID: 38686113
- PMCID: PMC11057235
- DOI: 10.3389/fmicb.2024.1376579
Pre-soil fumigation with ammonium bicarbonate and lime modulates the rhizosphere microbiome to mitigate clubroot disease in Chinese cabbage
Abstract
Background: Plasmodiophora brassicae is an ever-increasing threat to cruciferous crop production worldwide.
Aims and methods: This study investigated the impact of pre-soil fumigation with ammonium bicarbonate (N) and lime (NB) to manage clubroot disease in Chinese cabbage through 16S rRNA gene amplification sequencing.
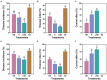
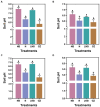
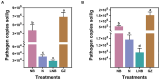
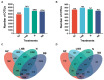
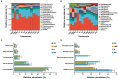
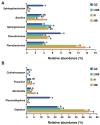
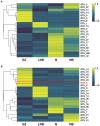
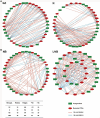
Results: We found that soil fumigation with N and NB suppressed disease incidence by reducing the soil acidity and population of P. brassicae in the rhizosphere. Minimum disease incidence and maximum relative control effect of about 74.68 and 66.28% were achieved in greenhouse and field experiments, respectively, under the combined application of ammonium bicarbonate and lime (LNB) as compared with N, NB, and control (GZ). Microbial diversity analysis through Miseq sequencing proved that pre-soil fumigation with N, NB, and LNB clearly manipulated rhizosphere microbial community composition and changed the diversity and structure of rhizosphere microbes compared with GZ. Bacterial phyla such as Proteobacteria, Bacteriodetes, and Acidobacteria and fungal phyla including Olpidiomycota and Ascomycota were most dominant in the rhizosphere of Chinese cabbage plants. Soil fumigation with N and NB significantly reduced the abundance of clubroot pathogen at genus (Plasmodiophora) level compared with GZ, while decreased further under combined application LNB. Microbial co-occurrence network analysis showed a highly connected and complex network and less competition for resources among microbes under combined application LNB.
Conclusion: We conclude that for environmentally friendly and sustainable agriculture, soil fumigation with combined ammonium bicarbonate and lime plays a crucial role in mitigating Chinese cabbage clubroot disease by alleviating soil pH, reducing pathogen population, and manipulating the rhizosphere microbiome.
Keywords: Plasmodiophora brassicae; biological control; disease suppression; rhizosphere microbiome; soil pH.
Copyright © 2024 Zhang, Zhou, Zhang, Dai, He, Qiu, Alharbi, Wei, Wei, Ahmed and Ji.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Crop Rotation with Marigold Promotes Soil Bacterial Structure to Assist in Mitigating Clubroot Incidence in Chinese Cabbage.Plants (Basel). 2022 Sep 2;11(17):2295. doi: 10.3390/plants11172295. Plants (Basel). 2022. PMID: 36079677 Free PMC article.
-
Microbial Consortia: An Engineering Tool to Suppress Clubroot of Chinese Cabbage by Changing the Rhizosphere Bacterial Community Composition.Biology (Basel). 2022 Jun 15;11(6):918. doi: 10.3390/biology11060918. Biology (Basel). 2022. PMID: 35741438 Free PMC article.
-
Lime and ammonium carbonate fumigation coupled with bio-organic fertilizer application steered banana rhizosphere to assemble a unique microbiome against Panama disease.Microb Biotechnol. 2019 May;12(3):515-527. doi: 10.1111/1751-7915.13391. Epub 2019 Mar 5. Microb Biotechnol. 2019. PMID: 30838803 Free PMC article.
-
Control Strategies of Clubroot Disease Caused by Plasmodiophora brassicae.Microorganisms. 2022 Mar 14;10(3):620. doi: 10.3390/microorganisms10030620. Microorganisms. 2022. PMID: 35336194 Free PMC article. Review.
-
Plasmodiophora brassicae: a review of an emerging pathogen of the Canadian canola (Brassica napus) crop.Mol Plant Pathol. 2012 Feb;13(2):105-13. doi: 10.1111/j.1364-3703.2011.00729.x. Epub 2011 Jun 1. Mol Plant Pathol. 2012. PMID: 21726396 Free PMC article. Review.
Cited by
-
Transcriptome profiling reveals the impact of various levels of biochar application on the growth of flue-cured tobacco plants.BMC Plant Biol. 2024 Jul 10;24(1):655. doi: 10.1186/s12870-024-05321-z. BMC Plant Biol. 2024. PMID: 38987695 Free PMC article.
-
Evaluation the role of Luteibacter pinisoli DP2-30 in mitigating pine wilt disease caused by Bursaphelenchus xylophilus through modulation of host microbiome.Front Plant Sci. 2025 Mar 5;16:1515506. doi: 10.3389/fpls.2025.1515506. eCollection 2025. Front Plant Sci. 2025. PMID: 40110359 Free PMC article.
-
Comparison of Root Transcriptomes against Clubroot Disease Pathogens in a Resistant Chinese Cabbage Cultivar (Brassica rapa cv. 'Akimeki').Plants (Basel). 2024 Aug 5;13(15):2167. doi: 10.3390/plants13152167. Plants (Basel). 2024. PMID: 39124284 Free PMC article.
-
Comprehensive review of Plasmodiophora brassicae: pathogenesis, pathotype diversity, and integrated control methods.Front Microbiol. 2025 Feb 6;16:1531393. doi: 10.3389/fmicb.2025.1531393. eCollection 2025. Front Microbiol. 2025. PMID: 39980695 Free PMC article. Review.
-
Unraveling the Mechanism of the Endophytic Bacterial Strain Pseudomonas oryzihabitans GDW1 in Enhancing Tomato Plant Growth Through Modulation of the Host Transcriptome and Bacteriome.Int J Mol Sci. 2025 Feb 23;26(5):1922. doi: 10.3390/ijms26051922. Int J Mol Sci. 2025. PMID: 40076548 Free PMC article.
References
-
- Ahmed W., Dai Z., Zhang J., Li S., Ahmed A., Munir S., et al. . (2022). Plant-microbe interaction: mining the impact of native Bacillus amyloliquefaciens WS-10 on tobacco bacterial wilt disease and rhizosphere microbial communities. Microbiol. Spectr. 10, e01471–e01422. doi: 10.1128/spectrum.01471-22 - DOI - PMC - PubMed
-
- Ahmed W., Dai Z., Zhang J., Shakeel Q., Kamaruzzaman M., Nosheen S., et al. . (2024). Ralstonia solanacearum differentially modulates soil physicochemical properties and rhizospheric bacteriome of resistant and susceptible tobacco cultivars. Microbiol. Res. 281:127604. doi: 10.1016/j.micres.2024.127604, PMID: - DOI - PubMed
-
- Anderson M. J., Walsh D. C. (2013). PERMANOVA, ANOSIM, and the mantel test in the face of heterogeneous dispersions: what null hypothesis are you testing? Ecol. Monogr. 83, 557–574. doi: 10.1890/12-2010.1 - DOI
-
- Bhattacharya I., Dutta S., Mondal S., Mondal B. J. C. J. O. P. P. (2014). Clubroot disease on Brassica crops in India. Can. J. Plant Pathol. 36, 154–160. doi: 10.1080/07060661.2013.875064 - DOI
LinkOut - more resources
Full Text Sources
Research Materials

