Targeting the postsynaptic scaffolding protein PSD-95 enhances BDNF signaling to mitigate depression-like behaviors in mice
- PMID: 38687826
- PMCID: PMC11223518
- DOI: 10.1126/scisignal.adn4556
Targeting the postsynaptic scaffolding protein PSD-95 enhances BDNF signaling to mitigate depression-like behaviors in mice
Abstract
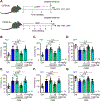
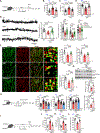
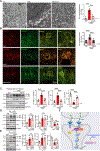
Signaling mediated by brain-derived neurotrophic factor (BDNF), which is supported by the postsynaptic scaffolding protein PSD-95, has antidepressant effects. Conversely, clinical depression is associated with reduced BDNF signaling. We found that peptidomimetic compounds that bind to PSD-95 promoted signaling by the BDNF receptor TrkB in the hippocampus and reduced depression-like behaviors in mice. The compounds CN2097 and Syn3 both bind to the PDZ3 domain of PSD-95, and Syn3 also binds to an α-helical region of the protein. Syn3 reduced depression-like behaviors in two mouse models of stress-induced depression; CN2097 had similar but less potent effects. In hippocampal neurons, application of Syn3 enhanced the formation of TrkB-Gαi1/3-PSD-95 complexes and potentiated downstream PI3K-Akt-mTOR signaling. In mice subjected to chronic mild stress (CMS), systemic administration of Syn3 reversed the CMS-induced, depression-associated changes in PI3K-Akt-mTOR signaling, dendrite complexity, spine density, and autophagy in the hippocampus and reduced depression-like behaviors. Knocking out Gαi1/3 in hippocampal neurons prevented the therapeutic effects of Syn3, indicating dependence of these effects on the TrkB pathway. The findings suggest that compounds that induce the formation of PSD-95-TrkB complexes have therapeutic potential to alleviate depression.
Conflict of interest statement
Figures





References
-
- Gill H, Gill B, Rodrigues NB, Lipsitz O, Rosenblat JD, El-Halabi S, Nasri F, Mansur RB, Lee Y, McIntyre RS, The Effects of Ketamine on Cognition in Treatment-Resistant Depression: A Systematic Review and Priority Avenues for Future Research. Neurosci Biobehav Rev 120, 78–85 (2021). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

