Predictive performance of pharmacokinetic-guided prophylactic dosing of factor concentrates in hemophilia A and B
- PMID: 38689619
- PMCID: PMC11058079
- DOI: 10.1016/j.rpth.2024.102397
Predictive performance of pharmacokinetic-guided prophylactic dosing of factor concentrates in hemophilia A and B
Abstract
Background: Pharmacokinetic (PK)-guided dosing is used to individualize factor (F)VIII and FIX replacement therapy.
Objectives: This study investigates the reliability and feasibility of PK-guided prophylactic dosing of factor concentrates in hemophilia A and B.
Methods: In this multicenter, prospective cohort study, people of all ages with hemophilia received prophylactic treatment with factor concentrates based on individual PK parameters. During follow-up, at least 4 measured FVIII/FIX levels per patient were compared with corresponding predicted levels obtained by Bayesian forecasting. Predictive performance was defined as adequate when ≥80% of measured FVIII/FIX levels were within ±25% of prediction (relative error). Additionally, mean absolute error and mean error were calculated. In post hoc analyses, predictive performance was assessed allowing maximum absolute errors of 1 (trough), 5 (mid), and 15 (peak) IU/dL. Five-point scale questionnaires addressed feasibility of PK guidance.
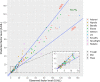
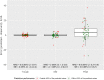
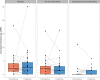
Results: We included 50 patients (median age, 19 years; range: 2-72 years). Median follow-up was 36 weeks. Seventy-one percent of levels (58% trough, 83% mid, and 80% peak) were within ±25% of prediction. Mean absolute errors were 0.8 (trough), 2.0 (mid), and 8.6 (peak) IU/dL. In post hoc analyses, 81% (trough), 96% (mid), and 82% (peak) of levels were within set limits. Patients reported low burden and high satisfaction.
Conclusion: PK-guided dosing was reliable according to post hoc analyses, based on low absolute errors that were regarded as clinically irrelevant in most cases. The predefined predictive performance was achieved in mid and peak factor levels but not in trough factor levels due to measurement inaccuracy. PK guidance also seemed feasible.
Keywords: factor IX; factor VIII; hemophilia A; hemophilia B; pharmacokinetics; preventive medicine.
© 2024 The Authors.
Figures


References
-
- Ahlberg A. Haemophilia in Sweden. VII. Incidence, treatment and prophylaxis of arthropathy and other musculo-skeletal manifestations of haemophilia A and B. Acta Orthop Scand Suppl. 1965;36(Suppl 77):3–132. - PubMed
-
- Manco-Johnson M.J., Abshire T.C., Shapiro A.D., Riske B., Hacker M.R., Kilcoyne R., et al. Prophylaxis versus episodic treatment to prevent joint disease in boys with severe hemophilia. N Engl J Med. 2007;357:535–544. - PubMed
-
- Björkman S., Folkesson A., Jönsson S. Pharmacokinetics and dose requirements of factor VIII over the age range 3-74 years: a population analysis based on 50 patients with long-term prophylactic treatment for haemophilia A. Eur J Clin Pharmacol. 2009;65:989–998. - PubMed
-
- Fijnvandraat K., Peters M., ten Cate J.W. Inter-individual variation in half-life of infused recombinant factor VIII is related to pre-infusion von Willebrand factor antigen levels. Br J Haematol. 1995;91:474–476. - PubMed
-
- Björkman S., Blanchette V.S., Fischer K., Oh M., Spotts G., Schroth P., et al. Comparative pharmacokinetics of plasma- and albumin-free recombinant factor VIII in children and adults: the influence of blood sampling schedule on observed age-related differences and implications for dose tailoring. J Thromb Haemost. 2010;8:730–736. - PMC - PubMed
LinkOut - more resources
Full Text Sources

