The clinical regimens and cell membrane camouflaged nanodrug delivery systems in hematologic malignancies treatment
- PMID: 38689664
- PMCID: PMC11059051
- DOI: 10.3389/fphar.2024.1376955
The clinical regimens and cell membrane camouflaged nanodrug delivery systems in hematologic malignancies treatment
Abstract
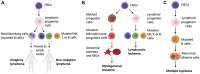
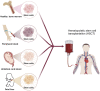
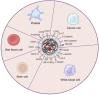
Hematologic malignancies (HMs), also referred to as hematological or blood cancers, pose significant threats to patients as they impact the blood, bone marrow, and lymphatic system. Despite significant clinical strategies using chemotherapy, radiotherapy, stem cell transplantation, targeted molecular therapy, or immunotherapy, the five-year overall survival of patients with HMs is still low. Fortunately, recent studies demonstrate that the nanodrug delivery system holds the potential to address these challenges and foster effective anti-HMs with precise treatment. In particular, cell membrane camouflaged nanodrug offers enhanced drug targeting, reduced toxicity and side effects, and/or improved immune response to HMs. This review firstly introduces the merits and demerits of clinical strategies in HMs treatment, and then summarizes the types, advantages, and disadvantages of current nanocarriers helping drug delivery in HMs treatment. Furthermore, the types, functions, and mechanisms of cell membrane fragments that help nanodrugs specifically targeted to and accumulate in HM lesions are introduced in detail. Finally, suggestions are given about their clinical translation and future designs on the surface of nanodrugs with multiple functions to improve therapeutic efficiency for cancers.
Keywords: cell membrane camouflage; clinical regimens; hematologic malignancies; nanocarrier; nanodrug delivery system.
Copyright © 2024 Liu, Yu, Chen, Hu, Fan and Liang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Recent advances of nanodrug delivery system in the treatment of hematologic malignancies.Semin Cancer Biol. 2022 Nov;86(Pt 2):607-623. doi: 10.1016/j.semcancer.2022.03.016. Epub 2022 Mar 23. Semin Cancer Biol. 2022. PMID: 35339668 Review.
-
Development and application of nanomaterials, nanotechnology and nanomedicine for treating hematological malignancies.J Hematol Oncol. 2023 Jun 23;16(1):65. doi: 10.1186/s13045-023-01460-2. J Hematol Oncol. 2023. PMID: 37353849 Free PMC article. Review.
-
Combined Strategies for Nanodrugs Noninvasively Overcoming the Blood-Brain Barrier and Actively Targeting Glioma Lesions.Biomater Res. 2025 Feb 5;29:0133. doi: 10.34133/bmr.0133. eCollection 2025. Biomater Res. 2025. PMID: 39911305 Free PMC article. Review.
-
Nanodrug Delivery Systems in Antitumor Immunotherapy.Biomater Res. 2024 Apr 25;28:0015. doi: 10.34133/bmr.0015. eCollection 2024. Biomater Res. 2024. PMID: 38840653 Free PMC article. Review.
-
Polymeric nanomedicines targeting hematological malignancies.J Control Release. 2021 Sep 10;337:571-588. doi: 10.1016/j.jconrel.2021.08.001. Epub 2021 Aug 6. J Control Release. 2021. PMID: 34364920 Review.
Cited by
-
Comparative Analysis of Clinical Outcomes and Financial Aspects of Phototherapies and Immunotherapy for Cancer.Adv Sci (Weinh). 2025 Aug;12(30):e17657. doi: 10.1002/advs.202417657. Epub 2025 Jun 19. Adv Sci (Weinh). 2025. PMID: 40536322 Free PMC article. Review.
References
-
- Abuasab T., Garcia-Manero G., Short N., Alvarado Y., Issa G. C., Islam R., et al. (2022). Phase 2 study of ASTX727 (cedazuridine/decitabine) plus venetoclax in patients with relapsed/refractory acute myeloid leukemia (AML) or previously untreated, elderly patients with AML unfit for chemotherapy. Blood 140 (Suppl. 1), 3324–3326. 10.1182/blood-2022-158566 - DOI
Publication types
LinkOut - more resources
Full Text Sources

