D-peptide-magnetic nanoparticles fragment tau fibrils and rescue behavioral deficits in a mouse model of Alzheimer's disease
- PMID: 38691615
- PMCID: PMC11062580
- DOI: 10.1126/sciadv.adl2991
D-peptide-magnetic nanoparticles fragment tau fibrils and rescue behavioral deficits in a mouse model of Alzheimer's disease
Abstract
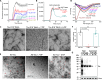
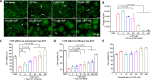
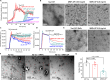
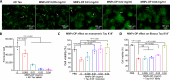
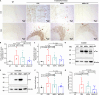
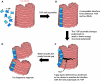
Amyloid fibrils of tau are increasingly accepted as a cause of neuronal death and brain atrophy in Alzheimer's disease (AD). Diminishing tau aggregation is a promising strategy in the search for efficacious AD therapeutics. Previously, our laboratory designed a six-residue, nonnatural amino acid inhibitor D-TLKIVW peptide (6-DP), which can prevent tau aggregation in vitro. However, it cannot block cell-to-cell transmission of tau aggregation. Here, we find D-TLKIVWC (7-DP), a d-cysteine extension of 6-DP, not only prevents tau aggregation but also fragments tau fibrils extracted from AD brains to neutralize their seeding ability and protect neuronal cells from tau-induced toxicity. To facilitate the transport of 7-DP across the blood-brain barrier, we conjugated it to magnetic nanoparticles (MNPs). The MNPs-DP complex retains the inhibition and fragmentation properties of 7-DP alone. Ten weeks of MNPs-DP treatment appear to reverse neurological deficits in the PS19 mouse model of AD. This work offers a direction for development of therapies to target tau fibrils.
Figures

References
-
- Braak H., Braak E., Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 82, 239–259 (1991). - PubMed
-
- Arriagada P. V., Growdon J. H., Hedley-Whyte E. T., Hyman B. T., Neurofibrillary tangles but not senile plaques parallel duration and severity of Alzheimer's disease. Neurology 42, 631–639 (1992). - PubMed
-
- Asher S., Priefer R., Alzheimer's disease failed clinical trials. Life Sci. 306, 120861 (2022). - PubMed
-
- Mullard A., Anti-tau antibody failures stack up. Nat. Rev. Drug Discov. 20, 888 (2021). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

