Promotive effect of skin precursor-derived Schwann cells on brachial plexus neurotomy and motor neuron damage repair through milieu-regulating secretome
- PMID: 38694448
- PMCID: PMC11061650
- DOI: 10.1016/j.reth.2024.04.002
Promotive effect of skin precursor-derived Schwann cells on brachial plexus neurotomy and motor neuron damage repair through milieu-regulating secretome
Abstract
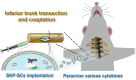
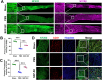
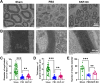
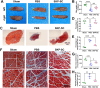
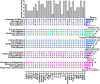
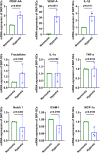
Brachial plexus injury (BPI) with motor neurons (MNs) damage still remain poor recovery in preclinical research and clinical therapy, while cell-based therapy approaches emerged as novel strategies. Previous work of rat skin precursor-derived Schwann cells (SKP-SCs) provided substantial foundation for repairing peripheral nerve injury (PNI). Given that, our present work focused on exploring the repair efficacy and possible mechanisms of SKP-SCs implantation on rat BPI combined with neurorrhaphy post-neurotomy. Results indicated the significant locomotive and sensory function recovery, with improved morphological remodeling of regenerated nerves and angiogenesis, as well as amelioration of target muscles atrophy and motor endplate degeneration. Besides, MNs could restore from oxygen-glucose-deprivation (OGD) injury upon SKP-SCs-sourced secretome treatment, implying the underlying paracrine mechanisms. Moreover, rat cytokine array assay detected 67 cytokines from SKP-SC-secretome, and bioinformatic analyses of screened 32 cytokines presented multiple functional clusters covering diverse cell types, including inflammatory cells, Schwann cells, vascular endothelial cells (VECs), neurons, and SKP-SCs themselves, relating distinct biological processes to nerve regeneration. Especially, a panel of hypoxia-responsive cytokines (HRCK), can participate into multicellular biological process regulation for permissive regeneration milieu, which underscored the benefits of SKP-SCs and sourced secretome, facilitating the chorus of nerve regenerative microenvironment. Furthermore, platelet-derived growth factor-AA (PDGF-AA) and vascular endothelial growth factor-A (VEGF-A) were outstanding cytokines involved with nerve regenerative microenvironment regulating, with significantly elevated mRNA expression level in hypoxia-responsive SKP-SCs. Altogether, through recapitulating the implanted SKP-SCs and derived secretome as niche sensor and paracrine transmitters respectively, HRCK would be further excavated as molecular underpinning of the neural recuperative mechanizations for efficient cell therapy; meanwhile, the analysis paradigm in this study validated and anticipated the actions and mechanisms of SKP-SCs on traumatic BPI repair, and was beneficial to identify promising bioactive molecule cocktail and signaling targets for cell-free therapy strategy on neural repair and regeneration.
Keywords: Branchial plexus injury; Hypoxia-responsive cytokines; Motor neurons; Regenerative microenvironment; Secretome; Skin precursor-derived Schwann cells.
© 2024, The Japanese Society for Regenerative Medicine. Production and hosting by Elsevier B.V.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Kaiser R., Waldauf P., Ullas G., Krajcova A. Epidemiology, etiology, and types of severe adult brachial plexus injuries requiring surgical repair: systematic review and meta-analysis. Neurosurg Rev. 2020;43:443–452. - PubMed
-
- Pejkova S., Filipce V., Peev I., Nikolovska B., Jovanoski T., Georgieva G., et al. Brachial plexus injuries - review of the anatomy and the treatment options. Prilozi. 2021;42:91–103. - PubMed
-
- Lauretti L., Pallini R., Romani R., Di Rocco F., Ciampini A., Gangitano C., et al. Lower trunk of brachial plexus injury in the neonate rat: effects of timing repair. Neurol Res. 2009;31:518–527. - PubMed
LinkOut - more resources
Full Text Sources

