Fentanyl activates opposing opioid and non-opioid receptor systems that control breathing
- PMID: 38698814
- PMCID: PMC11063261
- DOI: 10.3389/fphar.2024.1381073
Fentanyl activates opposing opioid and non-opioid receptor systems that control breathing
Abstract
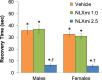
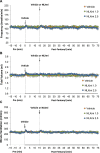
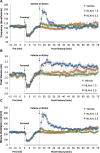
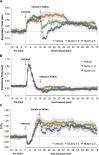
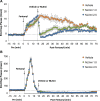
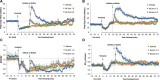
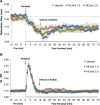
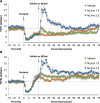
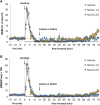
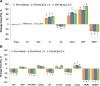
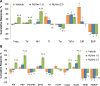
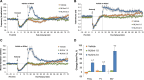
Fentanyl elicits profound disturbances in ventilatory control processes in humans and experimental animals. The traditional viewpoint with respect to fentanyl-induced respiratory depression is that once the effects on the frequency of breathing (Freq), tidal volume (TV), and minute ventilation (MV = Freq × TV) are resolved, then depression of breathing is no longer a concern. The results of the present study challenge this concept with findings, as they reveal that while the apparent inhibitory effects of fentanyl (75 μg/kg, IV) on Freq, TV, and MV in adult male rats were fully resolved within 15 min, many other fentanyl-induced responses were in full effect, including opposing effects on respiratory timing parameters. For example, although the effects on Freq were resolved at 15 min, inspiratory duration (Ti) and end inspiratory pause (EIP) were elevated, whereas expiratory duration (Te) and end expiratory pause (EEP) were diminished. Since the effects of fentanyl on TV had subsided fully at 15 min, it would be expected that the administration of an opioid receptor (OR) antagonist would have minimal effects if the effects of fentanyl on this and other parameters had resolved. We now report that the intravenous injection of a 1.0 mg/kg dose of the peripherally restricted OR antagonist, methyl-naloxone (naloxone methiodide, NLXmi), did not elicit arousal but elicited some relatively minor changes in Freq, TV, MV, Te, and EEP but pronounced changes in Ti and EIP. In contrast, the injection of a 2.5 mg/kg dose of NLXmi elicited pronounced arousal and dramatic changes in many variables, including Freq, TV, and MV, which were not associated with increases in non-apneic breathing events such as apneas. The two compelling conclusions from this study are as follows: 1) the blockade of central ORs produced by the 2.5 mg/kg dose of NLXmi elicits pronounced increases in Freq, TV, and MV in rats in which the effects of fentanyl had apparently resolved, and 2) it is apparent that fentanyl had induced the activation of two systems with counter-balancing effects on Freq and TV: one being an opioid receptor inhibitory system and the other being a non-OR excitatory system.
Keywords: breathing; fentanyl; naloxone methiodide; opioid receptors; rats.
Copyright © 2024 Baby, May, Getsy, Coffee, Nakashe, Bates, Levine and Lewis.
Conflict of interest statement
Author SB was employed by Galleon Pharmaceuticals, Inc. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures
Similar articles
-
Reversal of morphine-induced respiratory depression with the µ1-opioid receptor antagonist naloxonazine engenders excitation and instability of breathing.Am J Physiol Lung Cell Mol Physiol. 2025 Jul 1;329(1):L97-L111. doi: 10.1152/ajplung.00045.2025. Epub 2025 May 14. Am J Physiol Lung Cell Mol Physiol. 2025. PMID: 40366705 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
The opioid receptor antagonist naloxonazine uncovers the pronounced ventilatory stimulant effects of fentanyl in freely-moving rats.Eur J Pharmacol. 2025 Jul 8:177925. doi: 10.1016/j.ejphar.2025.177925. Online ahead of print. Eur J Pharmacol. 2025. PMID: 40639478
-
Mu-opioid antagonists for opioid-induced bowel dysfunction in people with cancer and people receiving palliative care.Cochrane Database Syst Rev. 2018 Jun 5;6(6):CD006332. doi: 10.1002/14651858.CD006332.pub3. Cochrane Database Syst Rev. 2018. Update in: Cochrane Database Syst Rev. 2022 Sep 15;9:CD006332. doi: 10.1002/14651858.CD006332.pub4. PMID: 29869799 Free PMC article. Updated.
-
Intravenous magnesium sulphate and sotalol for prevention of atrial fibrillation after coronary artery bypass surgery: a systematic review and economic evaluation.Health Technol Assess. 2008 Jun;12(28):iii-iv, ix-95. doi: 10.3310/hta12280. Health Technol Assess. 2008. PMID: 18547499
Cited by
-
Comparison of fentanyl-induced brain oxygen responses following intravenous and intraperitoneal injections in rats.Neuropharmacology. 2025 Jun 15;271:110412. doi: 10.1016/j.neuropharm.2025.110412. Epub 2025 Mar 12. Neuropharmacology. 2025. PMID: 40086623
-
Xylazine Exacerbates Fentanyl-Induced Respiratory Depression and Bradycardia.bioRxiv [Preprint]. 2024 Aug 19:2024.08.16.608310. doi: 10.1101/2024.08.16.608310. bioRxiv. 2024. Update in: J Pharmacol Exp Ther. 2025 Jul;392(7):103616. doi: 10.1016/j.jpet.2025.103616. PMID: 39229079 Free PMC article. Updated. Preprint.
-
Reversal of morphine-induced respiratory depression with the µ1-opioid receptor antagonist naloxonazine engenders excitation and instability of breathing.Am J Physiol Lung Cell Mol Physiol. 2025 Jul 1;329(1):L97-L111. doi: 10.1152/ajplung.00045.2025. Epub 2025 May 14. Am J Physiol Lung Cell Mol Physiol. 2025. PMID: 40366705 Free PMC article.
-
Searching for Synthetic Opioid Rescue Agents. 2: Identification of an Ultra-Potent Synthetic Opioid Rescue Agent.J Med Chem. 2025 Jun 26;68(12):13057-13074. doi: 10.1021/acs.jmedchem.5c01108. Epub 2025 Jun 11. J Med Chem. 2025. PMID: 40499097
-
Hypoxic effects of heroin and fentanyl and their basic physiological mechanisms.Am J Physiol Lung Cell Mol Physiol. 2024 Dec 1;327(6):L930-L948. doi: 10.1152/ajplung.00251.2024. Epub 2024 Oct 15. Am J Physiol Lung Cell Mol Physiol. 2024. PMID: 39404797 Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

