Immunogenic cell death mediated TLR3/4-activated MSCs in U87 GBM cell line
- PMID: 38698968
- PMCID: PMC11064142
- DOI: 10.1016/j.heliyon.2024.e29858
Immunogenic cell death mediated TLR3/4-activated MSCs in U87 GBM cell line
Abstract
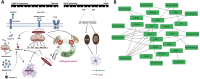
Background and aims: Glioblastoma (GBM) is an aggressive primary brain cancer with no promising curative therapies. It has been indicated that MSCs can interact with the tumour microenvironment (TME) through the secretion of soluble mediators regulating intercellular signalling within the TME. TLRs are a multigene family of pattern recognition receptors with evolutionarily conserved regions and are widely expressed in immune and other body cells. MSCs by TLRs can recognize conserved molecular components (DAPMPs and PAPMPs) and activate signalling pathways, which regulate immune and inflammatory responses. MSCs may exert immunomodulatory functions through interaction with their expressed toll-like receptors (TLRs) and exert a protective effect against tumour antigens. As an emerging approach, we aimed to monitor the U87 cell line growth, migration and death markers following specific TLR3/4-primed-MSCs-CMs treatment.
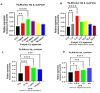
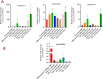
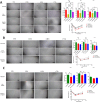
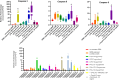
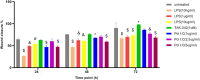
Methods and results: We investigated the phenotypic and functional outcomes of primed-CMs and glioma cell line co-culture following short-term, low-dose TLR3/4 priming. The gene expression profile of target genes, including apoptotic markers and related genes, was analyzed by qRT-PCR. MicroRNA-Seq examined the miRNA expression patterns, and flow cytometry evaluated the cell viability and cycle stages. The results showed significant changes in apoptosis and likely necroptosis-related markers following TLR3/4-primed-MSCs-CMs exposure in the glioma cell line. Notably, we observed a considerable induction of selective pro-apoptotic markers and both the early and late stages of apoptosis in treated U87 cell lines. Additionally, the migration rate of glioma cells significantly decreased following MSCs-CM treatment.
Conclusion: Our findings confirmed that the exposure of TLR3/4-activated-MSCs-CMs with glioma tumour cells possibly changes the immunogenicity of the tumour microenvironment and induces immunogenic programmed cell death. Our results can support the idea that TLR3/4-primed-MSCs can lead to innate immune-mediated cell death and modify tumour cell biology in invasive and metastatic cancers.
Keywords: Cell death; Mesenchymal stem cells; Priming; TLR3; TLR4; miRSeq.
© 2024 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





Similar articles
-
Mesenchymal Stromal Cells Primed by Toll-like Receptors 3 and 4 Enhanced Anti-Inflammatory Effects against LPS-Induced Macrophages via Extracellular Vesicles.Int J Mol Sci. 2023 Nov 13;24(22):16264. doi: 10.3390/ijms242216264. Int J Mol Sci. 2023. PMID: 38003458 Free PMC article.
-
The immunomodulatory function of equine MSCs is enhanced by priming through an inflammatory microenvironment or TLR3 ligand.Vet Immunol Immunopathol. 2018 Jan;195:33-39. doi: 10.1016/j.vetimm.2017.10.003. Epub 2017 Oct 31. Vet Immunol Immunopathol. 2018. PMID: 29249315
-
Differential effects of TLR3 and TLR4 activation on MSC-mediated immune regulation.Biochem Biophys Rep. 2024 Aug 10;39:101809. doi: 10.1016/j.bbrep.2024.101809. eCollection 2024 Sep. Biochem Biophys Rep. 2024. PMID: 39228386 Free PMC article.
-
Mesenchymal stromal cells (MSCs) and colorectal cancer: a troublesome twosome for the anti-tumour immune response?Oncotarget. 2016 Sep 13;7(37):60752-60774. doi: 10.18632/oncotarget.11354. Oncotarget. 2016. PMID: 27542276 Free PMC article. Review.
-
Cytokines play a key role in communication between mesenchymal stem cells and brain cancer cells.Protein Pept Lett. 2015;22(4):322-31. doi: 10.2174/0929866522666150131123808. Protein Pept Lett. 2015. PMID: 25642990 Review.
Cited by
-
Mesenchymal stromal cells in bone marrow niche of patients with multiple myeloma: a double-edged sword.Cancer Cell Int. 2025 Mar 26;25(1):117. doi: 10.1186/s12935-025-03741-x. Cancer Cell Int. 2025. PMID: 40140850 Free PMC article. Review.
References
-
- Lai Y., Lu X., Liao Y., Ouyang P., Wang H., Zhang X., et al. Crosstalk between glioblastoma and tumor microenvironment drives proneural–mesenchymal transition through ligand-receptor interactions. Genes Dis [Internet] 2024 Mar 1;11(2):874. [cited 2024 Mar 22] Available from:/pmc/articles/PMC10491977/ - PMC - PubMed
-
- Wu W., Klockow J., Zhang M., Fl-P . Elsevier [Internet]; 2021. Undefined. Glioblastoma Multiforme (GBM): an Overview of Current Therapies and Mechanisms of Resistance.https://www.sciencedirect.com/science/article/pii/S1043661821003649 [cited 2023 Jan 5]; Available from: - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

