Spatiotemporal Organization of Prefrontal Norepinephrine Influences Neuronal Activity
- PMID: 38702188
- PMCID: PMC11134306
- DOI: 10.1523/ENEURO.0252-23.2024
Spatiotemporal Organization of Prefrontal Norepinephrine Influences Neuronal Activity
Abstract
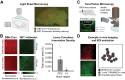
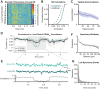
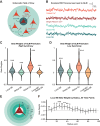
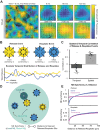
Norepinephrine (NE), a neuromodulator released by locus ceruleus (LC) neurons throughout the cortex, influences arousal and learning through extrasynaptic vesicle exocytosis. While NE within cortical regions has been viewed as a homogenous field, recent studies have demonstrated heterogeneous axonal dynamics and advances in GPCR-based fluorescent sensors permit direct observation of the local dynamics of NE at cellular scale. To investigate how the spatiotemporal dynamics of NE release in the prefrontal cortex (PFC) affect neuronal firing, we employed in vivo two-photon imaging of layer 2/3 of the PFC in order to observe fine-scale neuronal calcium and NE dynamics concurrently. In this proof of principle study, we found that local and global NE fields can decouple from one another, providing a substrate for local NE spatiotemporal activity patterns. Optic flow analysis revealed putative release and reuptake events which can occur at the same location, albeit at different times, indicating the potential to create a heterogeneous NE field. Utilizing generalized linear models, we demonstrated that cellular Ca2+ fluctuations are influenced by both the local and global NE field. However, during periods of local/global NE field decoupling, the local field drives cell firing dynamics rather than the global field. These findings underscore the significance of localized, phasic NE fluctuations for structuring cell firing, which may provide local neuromodulatory control of cortical activity.
Keywords: arousal; calcium imaging; computational modeling; norepinephrine; spatiotemporal activity; two-photon imaging.
Copyright © 2024 Glaeser-Khan et al.
Figures

Update of
-
Spatiotemporal organization of prefrontal norepinephrine influences neuronal activity.bioRxiv [Preprint]. 2023 Jul 19:2023.06.09.544191. doi: 10.1101/2023.06.09.544191. bioRxiv. 2023. Update in: eNeuro. 2024 May 28;11(5):ENEURO.0252-23.2024. doi: 10.1523/ENEURO.0252-23.2024. PMID: 37502881 Free PMC article. Updated. Preprint.
Similar articles
-
Spatiotemporal organization of prefrontal norepinephrine influences neuronal activity.bioRxiv [Preprint]. 2023 Jul 19:2023.06.09.544191. doi: 10.1101/2023.06.09.544191. bioRxiv. 2023. Update in: eNeuro. 2024 May 28;11(5):ENEURO.0252-23.2024. doi: 10.1523/ENEURO.0252-23.2024. PMID: 37502881 Free PMC article. Updated. Preprint.
-
Aging disrupts locus coeruleus-driven norepinephrine transmission in the prefrontal cortex: Implications for cognitive and motor decline.Aging Cell. 2025 Jan;24(1):e14342. doi: 10.1111/acel.14342. Epub 2024 Sep 23. Aging Cell. 2025. PMID: 39312903 Free PMC article.
-
Unilateral electrical stimulation of rat locus coeruleus elicits bilateral response of norepinephrine neurons and sustained activation of medial prefrontal cortex.J Neurophysiol. 2014 Jun 15;111(12):2570-88. doi: 10.1152/jn.00920.2013. Epub 2014 Mar 26. J Neurophysiol. 2014. PMID: 24671530
-
Synergistic tonic and phasic activity of the locus coeruleus norepinephrine (LC-NE) arousal system is required for optimal attentional performance.Metab Brain Dis. 2012 Sep;27(3):267-74. doi: 10.1007/s11011-012-9287-9. Epub 2012 Mar 8. Metab Brain Dis. 2012. PMID: 22399276 Review.
-
Norepinephrine versus dopamine and their interaction in modulating synaptic function in the prefrontal cortex.Brain Res. 2016 Jun 15;1641(Pt B):217-33. doi: 10.1016/j.brainres.2016.01.005. Epub 2016 Jan 11. Brain Res. 2016. PMID: 26790349 Free PMC article. Review.
Cited by
-
Innervation density governs crosstalk of GPCR-based norepinephrine and dopamine sensors.bioRxiv [Preprint]. 2024 Nov 23:2024.11.23.624963. doi: 10.1101/2024.11.23.624963. bioRxiv. 2024. PMID: 39605389 Free PMC article. Preprint.
-
Monoaminergic Modulation of Learning and Cognitive Function in the Prefrontal Cortex.Brain Sci. 2024 Sep 6;14(9):902. doi: 10.3390/brainsci14090902. Brain Sci. 2024. PMID: 39335398 Free PMC article. Review.
-
The Involvement of the Endocannabinoid, Glutamatergic, and GABAergic Systems in PTSD.Int J Mol Sci. 2025 Jun 20;26(13):5929. doi: 10.3390/ijms26135929. Int J Mol Sci. 2025. PMID: 40649707 Free PMC article. Review.
-
Frontal noradrenergic and cholinergic transients exhibit distinct spatiotemporal dynamics during competitive decision-making.Sci Adv. 2025 Mar 28;11(13):eadr9916. doi: 10.1126/sciadv.adr9916. Epub 2025 Mar 26. Sci Adv. 2025. PMID: 40138407 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
