Regulation of FSP1 myristoylation by NADPH: A novel mechanism for ferroptosis inhibition
- PMID: 38705094
- PMCID: PMC11074979
- DOI: 10.1016/j.redox.2024.103176
Regulation of FSP1 myristoylation by NADPH: A novel mechanism for ferroptosis inhibition
Abstract
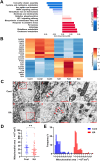
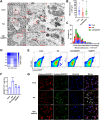
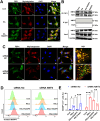
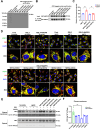
Excitotoxicity is a prevalent pathological event in neurodegenerative diseases. The involvement of ferroptosis in the pathogenesis of excitotoxicity remains elusive. Transcriptome analysis has revealed that cytoplasmic reduced nicotinamide adenine dinucleotide phosphate (NADPH) levels are associated with susceptibility to ferroptosis-inducing compounds. Here we show that exogenous NADPH, besides being reductant, interacts with N-myristoyltransferase 2 (NMT2) and upregulates the N-myristoylated ferroptosis suppressor protein 1 (FSP1). NADPH increases membrane-localized FSP1 and strengthens resistance to ferroptosis. Arg-291 of NMT2 is critical for the NADPH-NMT2-FSP1 axis-mediated suppression of ferroptosis. This study suggests that NMT2 plays a pivotal role by bridging NADPH levels and neuronal susceptibility to ferroptosis. We propose a mechanism by which the NADPH regulates N-myristoylation, which has important implications for ferroptosis and disease treatment.
Keywords: Excitotoxicity; FSP1; Ferroptosis; Myristoylation; NADPH; Neurodegenerative diseases.
Copyright © 2024 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures



References
-
- Liu Y., Nguyen M., Robert A., Meunier B. Metal ions in alzheimer's disease: a key role or not? Acc. Chem. Res. 2019;52:2026–2035. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

