Inhibition of Nogo-B reduces the progression of pancreatic cancer by regulation NF-κB/GLUT1 and SREBP1 pathways
- PMID: 38706871
- PMCID: PMC11068639
- DOI: 10.1016/j.isci.2024.109741
Inhibition of Nogo-B reduces the progression of pancreatic cancer by regulation NF-κB/GLUT1 and SREBP1 pathways
Abstract
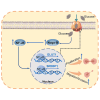
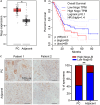
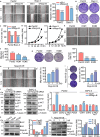
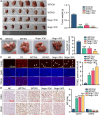
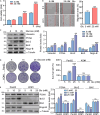
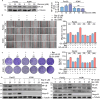
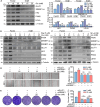
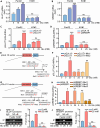
Pancreatic cancer (PC) is a lethal disease and associated with metabolism dysregulation. Nogo-B is related to multiple metabolic related diseases and types of cancers. However, the role of Nogo-B in PC remains unknown. In vitro, we showed that cell viability and migration was largely reduced in Nogo-B knockout or knockdown cells, while enhanced by Nogo-B overexpression. Consistently, orthotopic tumor and metastasis was reduced in global Nogo knockout mice. Furthermore, we indicated that glucose enhanced cell proliferation was associated to the elevation expression of Nogo-B and nuclear factor κB (NF-κB). While, NF-κB, glucose transporter type 1 (GLUT1) and sterol regulatory element-binding protein 1 (SREBP1) expression was reduced in Nogo-B deficiency cells. In addition, we showed that GLUT1 and SREBP1 was downstream target of NF-κB. Therefore, we demonstrated that Nogo deficiency inhibited PC progression is regulated by the NF-κB/GLUT1 and SREBP1 pathways, and suggested that Nogo-B may be a target for PC therapy.
Keywords: Cancer; Cell biology; Molecular biology.
© 2024 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
SREBP1-driven lipid desaturation supports clear cell renal cell carcinoma growth through regulation of NF-κB signaling.Biochem Biophys Res Commun. 2018 Jan 1;495(1):1383-1388. doi: 10.1016/j.bbrc.2017.11.163. Epub 2017 Nov 26. Biochem Biophys Res Commun. 2018. PMID: 29183723
-
SREBP1 promotes the invasion of colorectal cancer accompanied upregulation of MMP7 expression and NF-κB pathway activation.BMC Cancer. 2019 Jul 12;19(1):685. doi: 10.1186/s12885-019-5904-x. BMC Cancer. 2019. PMID: 31299935 Free PMC article.
-
An NF-κB/OVOL2 circuit regulates glucose import and cell survival in non-small cell lung cancer.Cell Commun Signal. 2022 Mar 28;20(1):40. doi: 10.1186/s12964-022-00845-z. Cell Commun Signal. 2022. PMID: 35346238 Free PMC article.
-
Downregulation of Nogo-B ameliorates cerebral ischemia/reperfusion injury in mice through regulating microglia polarization via TLR4/NF-kappaB pathway.Neurochem Int. 2023 Jul;167:105553. doi: 10.1016/j.neuint.2023.105553. Epub 2023 May 23. Neurochem Int. 2023. PMID: 37230196 Review.
-
The roles and mechanisms of SREBP1 in cancer development and drug response.Genes Dis. 2023 Jun 22;11(4):100987. doi: 10.1016/j.gendis.2023.04.022. eCollection 2024 Jul. Genes Dis. 2023. PMID: 38560498 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

