Deep Electroreductive Chemistry: Harnessing Carbon- and Silicon-based Reactive Intermediates in Organic Synthesis
- PMID: 38707967
- PMCID: PMC11067979
- DOI: 10.1021/acscatal.3c01174
Deep Electroreductive Chemistry: Harnessing Carbon- and Silicon-based Reactive Intermediates in Organic Synthesis
Abstract
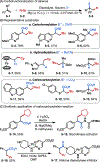
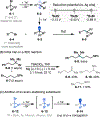
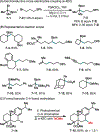
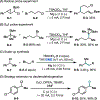
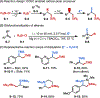
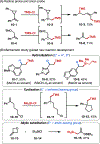
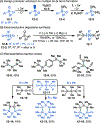
This Viewpoint outlines our recent contribution in electroreductive synthesis. Specifically, we leveraged deeply reducing potentials provided by electrochemistry to generate radical and anionic intermediates from readily available alkyl halides and chlorosilanes. Harnessing the distinct reactivities of radicals and anions, we have achieved several challenging transformations to construct C-C, C-Si, and Si-Si bonds. We highlight the mechanistic design principle that underpinned the development of each transformation and provide a view forward on future opportunities in growing area of reductive electrosynthesis.
Keywords: alkene difunctionalization; alkyl halide; chlorosilane; cross-electrophile coupling; electroreduction; electrosynthesis; radical-polar crossover.
Conflict of interest statement
The authors declare no competing financial interest.
Figures





References
-
- Moss RA; Platz MS; Jones M Jr. Reactive Intermediate Chemistry, Wiley-VCH, Weinheim, 2015.
-
- Hartwig J. Organotransition Metal Chemistry: From Bonding to Catalysis. University Science Books, New York, 2010
-
- List B. Introduction: Organocatalysis. Chem. Rev 2007, 107, 5413–5415.
- MacMillan DWC The advent and development of organocatalysis. Nature 2008, 455, 304–308. - PubMed
-
- Prier CK; Rankic DA; MacMillan DWC Visible Light Photoredox Catalysis with Transition Metal Complexes: Applications in Organic Synthesis. Chem. Rev 2013, 113, 5322–5363. - PMC - PubMed
- Narayanam JMR; Stephenson CRJ Visible Light Photoredox Catalysis: Applications in Organic Synthesis. Chem. Soc. Rev 2011, 40, 102–113. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
