Two promising Bacillus-derived antifungal lipopeptide leads AF4 and AF5 and their combined effect with fluconazole on the in vitro Candida glabrata biofilms
- PMID: 38708082
- PMCID: PMC11066293
- DOI: 10.3389/fphar.2024.1334419
Two promising Bacillus-derived antifungal lipopeptide leads AF4 and AF5 and their combined effect with fluconazole on the in vitro Candida glabrata biofilms
Abstract
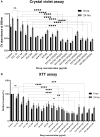
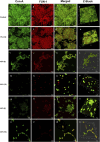
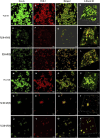
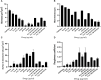
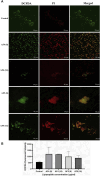
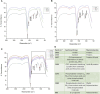
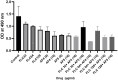
Introduction: Candida species are endowed with the ability to produce biofilms, which is one of the causes of pathogenicity, as biofilms protect yeasts from antifungal drugs. Candida glabrata (Nakaseomyces glabrata) is one of the most prevalent pathogenic yeasts in humans and a biofilm producer. Methods: The study was aimed at evaluating the combined effects of two highly promising antifungal biomolecules (AF4 and AF5) lipopeptide in nature, chromatographically purified to homogeneity from Bacillus subtilis (B. subtilis) and the standard antifungal fluconazole (at different concentrations) to demonstrate C. glabrata biofilm formation inhibition. Biofilm production and inhibition were evaluated by quantification of the biofilm biomass and metabolic activity using crystal violet (CV) staining and XTT reduction assays, respectively. Microscopic techniques such as confocal scanning laser microscopy (CSLM) and scanning electron microscopy (SEM) were employed to visualize biofilm formation and inhibition. Results and Discussion: Compared to untreated and fluconazole-treated biofilms, an enhanced in vitro anti-biofilm effect of the antifungal lipopeptides AF4/AF5 alone and their combinations with fluconazole was established. The lipopeptides AF4/AF5 alone at 8 and 16 μg/mL exhibited significant biomass and metabolic activity reductions. SEM and CSLM images provided evidence that the lipopeptide exposure results in architectural alterations and a significant reduction of C. glabrata biofilms, whereas (2', 7'-dichlorofluorescin diacetate (DCFDA) and propidium iodide (PI) analyses showed reactive oxygen species (ROS) generation along with membrane permeabilization. The estimation of exopolysaccharides (EPS) in AF4/AF5-treated biofilms indicated EPS reduction. The combinations of fluconazole (64/128 μg/mL) and AF4/AF5 lipopeptide (16 μg/mL) were found to significantly disrupt the mature (24 h) biofilms as revealed by CSLM and SEM studies. The CSLM images of biofilms were validated using COMSTAT. The FTIR-analyses indicate the antibiofilm effects of both lipopeptides on 24 h biofilms to support CSLM and SEM observations. The combinations of fluconazole (64/128 μg/mL) and AF4/AF5 lipopeptide were found to disrupt the mature biofilms; the study also showed that the lipopeptides alone have the potentials to combat C. glabrata biofilms. Taken together, it may be suggested that these lipopeptide leads can be optimized to potentially apply on various surfaces to either reduce or nearly eradicate yeast biofilms.
Keywords: Antifungal lipopeptide; Bacillus sp.; CV assay; Candida glabrata; XTT assay; biofilm inhibition; confocal microscopy.
Copyright © 2024 Madhuri, Rudramurthy and Roy.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Bacillus-derived lipopeptides inhibit and eradicate Candida tropicalis Biofilms: An in vitro exploration.Biofilm. 2025 Jun 14;10:100294. doi: 10.1016/j.bioflm.2025.100294. eCollection 2025 Dec. Biofilm. 2025. PMID: 40672400 Free PMC article.
-
Evaluation of Antifungal Efficacy of Three New Cyclic Lipopeptides of the Class Bacillomycin from Bacillus subtilis RLID 12.1.Antimicrob Agents Chemother. 2017 Dec 21;62(1):e01457-17. doi: 10.1128/AAC.01457-17. Print 2018 Jan. Antimicrob Agents Chemother. 2017. PMID: 29038271 Free PMC article.
-
Functional Characterization of a Bacillus-Derived Novel Broad-Spectrum Antifungal Lipopeptide Variant against Candida tropicalis and Candida auris and Unravelling Its Mode of Action.Microbiol Spectr. 2023 Feb 6;11(2):e0158322. doi: 10.1128/spectrum.01583-22. Online ahead of print. Microbiol Spectr. 2023. PMID: 36744953 Free PMC article.
-
Prevalence of biofilms in Candida spp. bloodstream infections: A meta-analysis.PLoS One. 2022 Feb 3;17(2):e0263522. doi: 10.1371/journal.pone.0263522. eCollection 2022. PLoS One. 2022. PMID: 35113972 Free PMC article. Review.
-
Candida Biofilms: Threats, Challenges, and Promising Strategies.Front Med (Lausanne). 2018 Feb 13;5:28. doi: 10.3389/fmed.2018.00028. eCollection 2018. Front Med (Lausanne). 2018. PMID: 29487851 Free PMC article. Review.
Cited by
-
Bacillus-derived lipopeptides inhibit and eradicate Candida tropicalis Biofilms: An in vitro exploration.Biofilm. 2025 Jun 14;10:100294. doi: 10.1016/j.bioflm.2025.100294. eCollection 2025 Dec. Biofilm. 2025. PMID: 40672400 Free PMC article.
References
-
- Ashley E. D., Johnson M. D. (2011). Combination antifungal therapy. Essentials Clin. Mycol. 48 (3), 153–163. 10.1007/978-1-4419-6640-7_10 - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

