Radiologic Lag and Brain MRI Lesion Dynamics During Attacks in MOG Antibody-Associated Disease
- PMID: 38710000
- PMCID: PMC11177594
- DOI: 10.1212/WNL.0000000000209303
Radiologic Lag and Brain MRI Lesion Dynamics During Attacks in MOG Antibody-Associated Disease
Abstract
Background and objectives: Knowledge of the evolution of CNS demyelinating lesions within attacks could assist diagnosis. We evaluated intra-attack lesion dynamics in patients with myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) vs multiple sclerosis (MS) and aquaporin-4 antibody seropositive neuromyelitis optica spectrum disorder (AQP4+NMOSD).
Methods: This retrospective observational multicenter study included consecutive patients from Mayo Clinic (USA) and Great Ormond Street Hospital for Children (UK). Inclusion criteria were as follows: (1) MOGAD, MS, or AQP4+NMOSD diagnosis; (2) availability of ≥2 brain MRIs (within 30 days of attack onset); and (3) brain involvement (i.e., ≥1 T2 lesion) on ≥1 brain MRI. The initial and subsequent brain MRIs within a single attack were evaluated for the following: new T2 lesions(s); resolved T2 lesion(s); both; or no change. This was compared between MOGAD, MS, and AQP4+NMOSD attacks. We used the Mann-Whitney U test and χ2/Fisher exact test for statistical analysis.
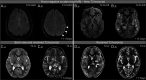
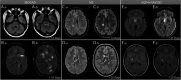
Results: Our cohort included 55 patients with MOGAD (median age, 14 years; interquartile range [IQR] 5-34; female sex, 29 [53%]) for a total of 58 attacks. The comparison groups included 38 patients with MS, and 19 with AQP4+NMOSD. In MOGAD, the initial brain MRI (median of 5 days from onset [IQR 3-9]) was normal in 6/58 (10%) attacks despite cerebral symptoms (i.e., radiologic lag). The commonest reason for repeat MRI was clinical worsening or no improvement (33/56 [59%] attacks with details available). When compared with the first MRI, the second intra-attack MRI (median of 8 days from initial scan [IQR 5-13]) showed the following: new T2 lesion(s) 27/58 (47%); stability 24/58 (41%); resolution of T2 lesion(s) 4/58 (7%); or both new and resolved T2 lesions 3/58 (5%). Findings were similar between children and adults. Steroid treatment was associated with resolution of ≥1 T2 lesion (6/28 [21%] vs 1/30 [3%], p = 0.048) and reduced the likelihood of new T2 lesions (9/28 vs 18/30, p = 0.03). Intra-attack MRI changes favored MOGAD (34/58 [59%]) over MS (10/38 [26%], p = 0.002) and AQP4+NMOSD (4/19 [21%], p = 0.007). Resolution of ≥1 T2 lesions was exclusive to MOGAD (7/58 [12%]).
Discussion: Radiologic lag is common within MOGAD attacks. Dynamic imaging with frequent appearance and occasional disappearance of lesions within a single attack suggest MOGAD diagnosis over MS and AQP4+NMOSD. These findings have implications for clinical practice, clinical trial attack adjudication, and understanding of MOGAD pathogenesis.
Conflict of interest statement
L. Cacciaguerra received speaker and consultant honoraria from ACCMED, Roche, BMS Celgene, and Sanofi and travel support for conferences from Merck-Serono. O. Abdel-Mannan receives funding from the Association of British Neurologists, MS Society, and the Berkeley Foundation. D. Champsas, K. Mankad, and K.N. Krecke report no disclosures. J.J. Chen reports payment for consultation from UCB, Horizon, and Roche. S.B. Syc-Mazurek and V. Redenbaugh report no disclosures. A.S. Lopez Chiriboga has served on advisory boards for Genentech and Horizon Therapeutics. C. Valencia-Sanchez reports no disclosures. C. Hemingway reports grant support from the MRC, MS Society and Vasculitis UK. She has served as a consultant to Novartis, Roche, UCB, and Sanofi. She has participated on various IDMCs. J.-M. Tillema is Associate Editor for
Figures


Similar articles
-
Frequency of New Silent MRI Lesions in Myelin Oligodendrocyte Glycoprotein Antibody Disease and Aquaporin-4 Antibody Neuromyelitis Optica Spectrum Disorder.JAMA Netw Open. 2021 Dec 1;4(12):e2137833. doi: 10.1001/jamanetworkopen.2021.37833. JAMA Netw Open. 2021. PMID: 34878547 Free PMC article.
-
Comparison of MRI T2-lesion evolution in pediatric MOGAD, NMOSD, and MS.Mult Scler. 2023 Jun;29(7):799-808. doi: 10.1177/13524585231166834. Epub 2023 May 23. Mult Scler. 2023. PMID: 37218499 Free PMC article.
-
Comparison of MRI Lesion Evolution in Different Central Nervous System Demyelinating Disorders.Neurology. 2021 Sep 14;97(11):e1097-e1109. doi: 10.1212/WNL.0000000000012467. Epub 2021 Jul 14. Neurology. 2021. PMID: 34261784 Free PMC article.
-
Characteristics of MRI lesions in AQP4 antibody-positive NMOSD, MOGAD, and multiple sclerosis: a systematic review and meta-analysis.J Neurol. 2025 Aug 7;272(9):560. doi: 10.1007/s00415-025-13303-w. J Neurol. 2025. PMID: 40773034 Free PMC article.
-
NMOSD and MOGAD.Continuum (Minneap Minn). 2024 Aug 1;30(4):1052-1087. doi: 10.1212/CON.0000000000001454. Continuum (Minneap Minn). 2024. PMID: 39088288 Review.
Cited by
-
Testing for myelin oligodendrocyte glycoprotein antibodies: Who, what, where, when, why, and how.Mult Scler. 2025 Apr;31(5):505-511. doi: 10.1177/13524585251313744. Epub 2025 Jan 24. Mult Scler. 2025. PMID: 39861933 Free PMC article. Review.
-
Alterations in peripheral blood immune cell profiles of neuromyelitis optica spectrum disorder across different phases and after B cell depletion therapy.Front Immunol. 2025 Jun 11;16:1556259. doi: 10.3389/fimmu.2025.1556259. eCollection 2025. Front Immunol. 2025. PMID: 40568579 Free PMC article.
-
Clinical Features of Children With MOG-IgG Who Fulfill Criteria of Multiple Sclerosis and Overlapping Disorders.Neurol Neuroimmunol Neuroinflamm. 2025 Jul;12(4):e200400. doi: 10.1212/NXI.0000000000200400. Epub 2025 Jun 12. Neurol Neuroimmunol Neuroinflamm. 2025. PMID: 40505071 Free PMC article.
-
Radiological features in pediatric myelin oligodendrocyte glycoprotein antibody-associated disease-diagnostic criteria and lesion dynamics.Pediatr Radiol. 2025 Apr;55(4):699-720. doi: 10.1007/s00247-024-06023-2. Epub 2024 Sep 7. Pediatr Radiol. 2025. PMID: 39243314 Free PMC article. Review.
-
Magnetic resonance imaging-based biomarkers of multiple sclerosis and neuromyelitis optica spectrum disorder: a systematic review and meta-analysis.J Neurol. 2024 Dec 16;272(1):77. doi: 10.1007/s00415-024-12827-x. J Neurol. 2024. PMID: 39680165
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
