Phosphorylation-Dependent Regulation of Guanylyl Cyclase (GC)-A and Other Membrane GC Receptors
- PMID: 38713083
- PMCID: PMC11405504
- DOI: 10.1210/endrev/bnae015
Phosphorylation-Dependent Regulation of Guanylyl Cyclase (GC)-A and Other Membrane GC Receptors
Abstract
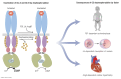
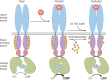
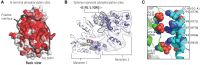
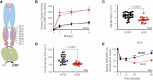
Receptor guanylyl cyclases (GCs) are single membrane spanning, multidomain enzymes, that synthesize cGMP in response to natriuretic peptides or other ligands. They are evolutionarily conserved from sea urchins to humans and regulate diverse physiologies. Most family members are phosphorylated on 4 to 7 conserved serines or threonines at the beginning of their kinase homology domains. This review describes studies that demonstrate that phosphorylation and dephosphorylation are required for activation and inactivation of these enzymes, respectively. Phosphorylation sites in GC-A, GC-B, GC-E, and sea urchin receptors are discussed, as are mutant receptors that mimic the dephosphorylated inactive or phosphorylated active forms of GC-A and GC-B, respectively. A salt bridge model is described that explains why phosphorylation is required for enzyme activation. Potential kinases, phosphatases, and ATP regulation of GC receptors are also discussed. Critically, knock-in mice with glutamate substitutions for receptor phosphorylation sites are described. The inability of opposing signaling pathways to inhibit cGMP synthesis in mice where GC-A or GC-B cannot be dephosphorylated demonstrates the necessity of receptor dephosphorylation in vivo. Cardiac hypertrophy, oocyte meiosis, long-bone growth/achondroplasia, and bone density are regulated by GC phosphorylation, but additional processes are likely to be identified in the future.
Keywords: Npr1; Npr2; achondroplasia; cardiac hypertrophy; osteoporosis.
© The Author(s) 2024. Published by Oxford University Press on behalf of the Endocrine Society. All rights reserved. For commercial re-use, please contact reprints@oup.com for reprints and translation rights for reprints. All other permissions can be obtained through our RightsLink service via the Permissions link on the article page on our site—for further information please contact journals.permissions@oup.com. See the journal About page for additional terms.
Figures


References
-
- Hardman JG, Sutherland EW. Guanyl cyclase, an enzyme catalyzing the formation of guanosine 3',5'-monophosphate from guanosine trihosphate. J Biol Chem. 1969;244(23):6363‐6370. - PubMed
-
- Ishikawa E, Ishikawa S, Davis JW, Sutherland EW. Determination of guanosine 3',5'-monophosphate in tissues and of guanyl cyclase in rat intestine. J Biol Chem. 1969;244(23):6371‐6376. - PubMed
-
- White AA, Aurbach GD. Detection of guanyl cyclase in mammalian tissues. Biochim Biophys Acta. 1969;191(3):686‐697. - PubMed
-
- Schultz G, Böhme E, Munske K. Guanyl cyclase. Determination of enzyme activity. Life Sci. 1969;8(24):1323‐1332. - PubMed
-
- Hansbrough JR, Garbers DL. Speract. Purification and characterization of a peptide associated with eggs that activates spermatozoa. J Biol Chem. 1981;256(3):1447‐1452. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

