Neurons upregulate PD-L1 via IFN/STAT1/IRF1 to alleviate damage by CD8+ T cells in cerebral malaria
- PMID: 38715061
- PMCID: PMC11077882
- DOI: 10.1186/s12974-024-03114-7
Neurons upregulate PD-L1 via IFN/STAT1/IRF1 to alleviate damage by CD8+ T cells in cerebral malaria
Abstract
Background: Cerebral malaria (CM) is the most lethal complication of malaria, and survivors usually endure neurological sequelae. Notably, the cytotoxic effect of infiltrating Plasmodium-activated CD8+ T cells on cerebral microvasculature endothelial cells is a prominent feature of the experimental CM (ECM) model with blood-brain barrier disruption. However, the damage effect of CD8+ T cells infiltrating the brain parenchyma on neurons remains unclear. Based on the immunosuppressive effect of the PD-1/PD-L1 pathway on T cells, our previous study demonstrated that the systemic upregulation of PD-L1 to inhibit CD8+ T cell function could effectively alleviate the symptoms of ECM mice. However, it has not been reported whether neurons can suppress the pathogenic effect of CD8+ T cells through the PD-1/PD-L1 negative immunomodulatory pathway. As the important inflammatory factor of CM, interferons can induce the expression of PD-L1 via different molecular mechanisms according to the neuro-immune microenvironment. Therefore, this study aimed to investigate the direct interaction between CD8+ T cells and neurons, as well as the mechanism of neurons to alleviate the pathogenic effect of CD8+ T cells through up-regulating PD-L1 induced by IFNs.
Methods: Using the ECM model of C57BL/6J mice infected with Plasmodium berghei ANKA (PbA), morphological observations were conducted in vivo by electron microscope and IF staining. The interaction between the ECM CD8+ T cells (immune magnetic bead sorting from spleen of ECM mice) and primary cultured cortical neurons in vitro was observed by IF staining and time-lapse photography. RNA-seq was performed to analyze the signaling pathway of PD-L1 upregulation in neurons induced by IFNβ or IFNγ, and verified through q-PCR, WB, IF staining, and flow cytometry both in vitro and in vivo using IFNAR or IFNGR gene knockout mice. The protective effect of adenovirus-mediated PD-L1 IgGFc fusion protein expression was verified in ECM mice with brain stereotaxic injection in vivo and in primary cultured neurons via viral infection in vitro.
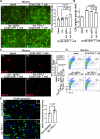
Results: In vivo, ECM mice showed infiltration of activated CD8+ T cells and neuronal injury in the brain parenchyma. In vitro, ECM CD8+ T cells were in direct contact with neurons and induced axonal damage, as an active behavior. The PD-L1 protein level was elevated in neurons of ECM mice and in primary cultured neurons induced by IFNβ, IFNγ, or ECM CD8+ T cells in vitro. Furthermore, the IFNβ or IFNγ induced neuronal expression of PD-L1 was mediated by increasing STAT1/IRF1 pathway via IFN receptors. The increase of PD-L1 expression in neurons during PbA infection was weakened after deleting the IFNAR or IFNGR. Increased PD-L1 expression by adenovirus partially protected neurons from CD8+ T cell-mediated damage both in vitro and in vivo.
Conclusion: Our study demonstrates that both type I and type II IFNs can induce neurons to upregulate PD-L1 via the STAT1/IRF1 pathway mediated by IFN receptors to protect against activated CD8+ T cell-mediated damage, providing a targeted pathway to alleviate neuroinflammation during ECM.
Keywords: CD8+ T cell; Experimental cerebral malaria; Interferon; Neuron; PD-L1.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures







References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

