Diabetic retinopathy is a ceramidopathy reversible by anti-ceramide immunotherapy
- PMID: 38718792
- PMCID: PMC11222062
- DOI: 10.1016/j.cmet.2024.04.013
Diabetic retinopathy is a ceramidopathy reversible by anti-ceramide immunotherapy
Abstract
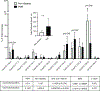
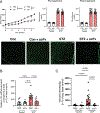
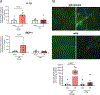
Diabetic retinopathy is a microvascular disease that causes blindness. Using acid sphingomyelinase knockout mice, we reported that ceramide generation is critical for diabetic retinopathy development. Here, in patients with proliferative diabetic retinopathy, we identify vitreous ceramide imbalance with pathologic long-chain C16-ceramides increasing and protective very long-chain C26-ceramides decreasing. C16-ceramides generate pro-inflammatory/pro-apoptotic ceramide-rich platforms on endothelial surfaces. To geo-localize ceramide-rich platforms, we invented a three-dimensional confocal assay and showed that retinopathy-producing cytokines TNFα and IL-1β induce ceramide-rich platform formation on retinal endothelial cells within seconds, with volumes increasing 2-logs, yielding apoptotic death. Anti-ceramide antibodies abolish these events. Furthermore, intravitreal and systemic anti-ceramide antibodies protect from diabetic retinopathy in standardized rodent ischemia reperfusion and streptozotocin models. These data support (1) retinal endothelial ceramide as a diabetic retinopathy treatment target, (2) early-stage therapy of non-proliferative diabetic retinopathy to prevent progression, and (3) systemic diabetic retinopathy treatment; and they characterize diabetic retinopathy as a "ceramidopathy" reversible by anti-ceramide immunotherapy.
Keywords: IL-1β; TNFα; apoptosis; ceramide; diabetes; diabetic retinopathy; endothelial cell; inflammation; retina; sphingomyelinase.
Copyright © 2024 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests R.N.K. is a founder of Ceramedix Holding LLC. Patents to R.N.K.: US7195775B1, US7850984B2, US10052387B2, US10414533B2, US20140205543A1, US8562993B2, US9592238B2, US10450385B2, US20150216971A1, US20170335014A1, US20190389970A1, US20190046538A1, US20170333413A1, and US20180015183A1. J.V.B. is a consultant for Ceramedix, Inc., subsidiary of Ceramedix Holding LLC. Patent to J.V.B.: US10975169B1.
Figures




References
-
- Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Mbanya JC, et al. (2022). IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract 183, 109119. 10.1016/j.diabres.2021.109119. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

