Predicting vaccine effectiveness for mpox
- PMID: 38719852
- PMCID: PMC11078999
- DOI: 10.1038/s41467-024-48180-w
Predicting vaccine effectiveness for mpox
Abstract
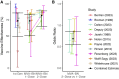
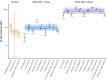
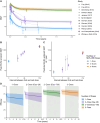
The Modified Vaccinia Ankara vaccine developed by Bavarian Nordic (MVA-BN) was widely deployed to prevent mpox during the 2022 global outbreak. This vaccine was initially approved for mpox based on its reported immunogenicity (from phase I/II trials) and effectiveness in animal models, rather than evidence of clinical efficacy. However, no validated correlate of protection after vaccination has been identified. Here we performed a systematic search and meta-analysis of the available data to test whether vaccinia-binding ELISA endpoint titer is predictive of vaccine effectiveness against mpox. We observe a significant correlation between vaccine effectiveness and vaccinia-binding antibody titers, consistent with the existing assumption that antibody levels may be a correlate of protection. Combining this data with analysis of antibody kinetics after vaccination, we predict the durability of protection after vaccination and the impact of dose spacing. We find that delaying the second dose of MVA-BN vaccination will provide more durable protection and may be optimal in an outbreak with limited vaccine stock. Although further work is required to validate this correlate, this study provides a quantitative evidence-based approach for using antibody measurements to predict the effectiveness of mpox vaccination.
© 2024. The Author(s).
Conflict of interest statement
C.R.M. is on the WHO SAGE Working Group on Smallpox and Monkeypox. The authors have no other competing interests to declare.
Figures

References
-
- WHO Emergency Response Team. Multi-country outbreak of mpox, External situation report#29. (2023). https://www.who.int/publications/m/item/multi-country-outbreak-of-mpox-e....
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

