Nitro-oleic acid (NO2-OA) ameliorates erectile dysfunction in a rat model of diabetes mellitus via modulation of fibrosis, inflammation and autophagy
- PMID: 38721285
- PMCID: PMC11074686
- DOI: 10.21037/tau-23-547
Nitro-oleic acid (NO2-OA) ameliorates erectile dysfunction in a rat model of diabetes mellitus via modulation of fibrosis, inflammation and autophagy
Abstract
Background: Inflammation, fibrosis and autophagy represent closely related factors associated with the pathogenesis of diabetes mellitus erectile dysfunction (DMED). In this study, the therapeutic effect of nitro-oleic acid (NO2-OA) in a streptozotocin-induced rat model of DMED was evaluated.
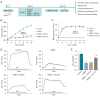
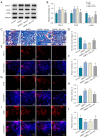
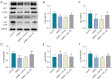
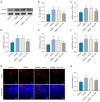
Methods: Sixty rats were randomly divided into four groups: control, DMED, DMED + Vehicle and DMED + NO2-OA. DMED was induced by intraperitoneal injection of streptozotocin in male rats. Blood glucose and body weight were measured every 2 weeks. After 4 weeks of NO2-OA treatment, erectile function was measured by electrical stimulation of cavernous nerve (CN). Western blotting, quantitative real-time polymerase chain reaction (qRT-PCR), enzyme-linked immunosorbent assay (ELISA), immunofluorescence and Masson's trichrome staining were used to verify the related factors and protein expression levels.
Results: We found that NO2-OA could significantly increase erectile pressure in the corpus cavernosum of DMED rats. Results of western blot, confocal immunofluorescence and qRT-PCR assays revealed that NO2-OA significantly reduced inflammatory factors and the expression of nuclear factor kappa B (NF-κB). In addition, Masson staining results indicated that NO2-OA significantly reduced the display of fibrotic tissue in the corpus cavernosum. These beneficial effects may be related to reductions in the expression of transforming growth factor-β1 (TGF-β1) and connective tissue growth factor (CTGF) and the increase in the expression of α-smooth muscle actin (α-SMA). Finally, NO2-OA treatment increased the expression of the autophagy marker, LC3, while P62 was decreased, effects suggesting that one of the underlying mechanisms of NO2-OA may involve an activation of the PI3K/AKT/mTOR pathway to enhance the capacity for autophagy within this tissue.
Conclusions: NO2-OA enhances erectile function within a rat model of DMED by inhibiting inflammation and fibrosis along with activating autophagy.
Keywords: Diabetes mellitus erectile dysfunction (DMED); autophagy; fibrosis; inflammation; nitro-oleic acid (NO2-OA).
2024 Translational Andrology and Urology. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tau.amegroups.com/article/view/10.21037/tau-23-547/coif). The authors have no conflicts of interest to declare.
Figures
References
LinkOut - more resources
Full Text Sources
Miscellaneous
