Sensory Stimulation-dependent Npas4 Expression in the Olfactory Bulb during Early Postnatal Development
- PMID: 38724478
- PMCID: PMC11089401
- DOI: 10.5607/en23037
Sensory Stimulation-dependent Npas4 Expression in the Olfactory Bulb during Early Postnatal Development
Abstract
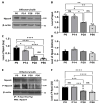
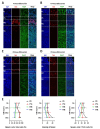
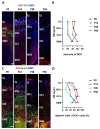
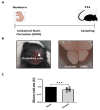
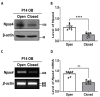
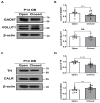
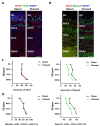
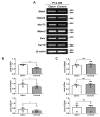
The development of the olfactory system is influenced by sensory inputs, and it maintains neuronal generation and plasticity throughout the lifespan. The olfactory bulb contains a higher proportion of interneurons than other brain regions, particularly during the early postnatal period of neurogenesis. Although the relationship between sensory stimulation and olfactory bulb development during the postnatal period has been well studied, the molecular mechanisms have yet to be identified. In this study, we used western blotting and immunohistochemistry to analyze the expression of the transcription factor Npas4, a neuron-specific immediate-early gene that acts as a developmental regulator in many brain regions. We found that Npas4 is highly expressed in olfactory bulb interneurons during the early postnatal stages and gradually decreases toward the late postnatal stages. Npas4 expression was observed in all olfactory bulb layers, including the rostral migratory stream, where newborn neurons are generated and migrate to the olfactory bulb. Under sensory deprivation, the olfactory bulb size and the number of olfactory bulb interneurons were reduced. Furthermore, Npas4 expression and the expression of putative Npas4 downstream molecules were decreased. Collectively, these findings indicate that Npas4 expression induced by sensory input plays a role in the formation of neural circuits with excitatory mitral/tufted cells by regulating the survival of olfactory bulb interneurons during the early stages of postnatal development.
Keywords: Immediate-early gene; Npas4; Olfactory bulb development; Olfactory bulb interneurons; Postnatal development; Unilateral naris occlusion.
Figures

Similar articles
-
Molecular Mechanisms Regulating the Dendritic Development of Newborn Olfactory Bulb Interneurons in a Sensory Experience-Dependent Manner.Front Neurosci. 2016 Jan 12;9:514. doi: 10.3389/fnins.2015.00514. eCollection 2015. Front Neurosci. 2016. PMID: 26793053 Free PMC article. Review.
-
Npas4 regulates Mdm2 and thus Dcx in experience-dependent dendritic spine development of newborn olfactory bulb interneurons.Cell Rep. 2014 Aug 7;8(3):843-57. doi: 10.1016/j.celrep.2014.06.056. Epub 2014 Jul 31. Cell Rep. 2014. PMID: 25088421
-
A subtype-specific critical period for neurogenesis in the postnatal development of mouse olfactory glomeruli.PLoS One. 2012;7(11):e48431. doi: 10.1371/journal.pone.0048431. Epub 2012 Nov 1. PLoS One. 2012. PMID: 23133633 Free PMC article.
-
Intrinsic Neuronal Activity during Migration Controls the Recruitment of Specific Interneuron Subtypes in the Postnatal Mouse Olfactory Bulb.J Neurosci. 2021 Mar 24;41(12):2630-2644. doi: 10.1523/JNEUROSCI.1960-20.2021. Epub 2021 Feb 3. J Neurosci. 2021. PMID: 33536198 Free PMC article.
-
Subpopulations of Projection Neurons in the Olfactory Bulb.Front Neural Circuits. 2020 Aug 28;14:561822. doi: 10.3389/fncir.2020.561822. eCollection 2020. Front Neural Circuits. 2020. PMID: 32982699 Free PMC article. Review.
Cited by
-
Urine miRNA signature as potential non-invasive diagnostic biomarker for Hirschsprung's disease.Front Mol Neurosci. 2025 Jan 8;17:1504424. doi: 10.3389/fnmol.2024.1504424. eCollection 2024. Front Mol Neurosci. 2025. PMID: 39872605 Free PMC article.

