The pan-PPAR agonist lanifibranor improves cardiometabolic health in patients with metabolic dysfunction-associated steatohepatitis
- PMID: 38730247
- PMCID: PMC11087475
- DOI: 10.1038/s41467-024-47919-9
The pan-PPAR agonist lanifibranor improves cardiometabolic health in patients with metabolic dysfunction-associated steatohepatitis
Abstract
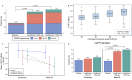
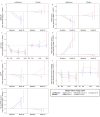
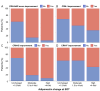
Lanifibranor, a pan-PPAR agonist, improves liver histology in patients with metabolic dysfunction-associated steatohepatitis (MASH), who have poor cardiometabolic health (CMH) and cardiovascular events as major mortality cause. NATIVE trial secondary and exploratory outcomes (ClinicalTrials.gov NCT03008070) were analyzed for the effect of lanifibranor on IR, lipid and glucose metabolism, systemic inflammation, blood pressure (BP), hepatic steatosis (imaging and histological grading) for all patients of the original analysis. With lanifibranor, triglycerides, HDL-C, apolipoproteins, insulin, HOMA-IR, HbA1c, fasting glucose (FG), hs-CRP, ferritin, diastolic BP and steatosis improved significantly, independent of diabetes status: most patients with prediabetes returned to normal FG levels. Significant adiponectin increases correlated with hepatic and CMH marker improvement; patients had an average weight gain of 2.5 kg, with 49% gaining ≥2.5% weight. Therapeutic benefits were similar regardless of weight change. Here, we show that effects of lanifibranor on liver histology in MASH are accompanied with CMH improvement, indicative of potential cardiovascular clinical benefits.
© 2024. The Author(s).
Conflict of interest statement
M.P.C., L.D., P.H.M., M.B. and P.B. are employees of Inventiva. J.L.J. is a consultant for Inventiva. D.R.B. has received honoraria from Inventiva via Translational Medicine Academy. J.B. has served as a consultant for Abbott, American Regent, Amgen, Applied Therapeutic, AskBio, Astellas, AstraZeneca, Bayer, Boehringer Ingelheim, Boston Scientific, Bristol Myers Squibb, Cardiac Dimension, Cardiocell, Cardior, Cardiorem, CSL Bearing, CVRx, Cytokinetics, Daxor, Edwards, Element Science, Faraday, Foundry, G3P, Innolife, Impulse Dynamics, Imbria, Inventiva, Ionis, Lexicon, Lilly, LivaNova, Janssen, Medtronics, Merck, Occlutech, Owkin, Novartis, Novo Nordisk, Pfizer, Pharmacosmos, Pharmain, Prolaio, Regeneron, Renibus, Roche, Salamandra, Sanofi, SC Pharma, Secretome, Sequana, SQ Innovation, Tenex, Tricog, Ultromics, Vifor, and Zoll. RPG received research support or honoraria form Amgen, Anthos Therapeutics, Daiichi Sankyo, Ionis, Dr. Reddy’s Laboratories, Medical Education Resources, Menarini, SAJA Pharmaceuticals, Servier, SUMMEET, and consults for Artivion, Beckman Coulter, Daiichi Sankyo, Gilead, Inari, Inventiva, PhaseBio Pharmaceuticals, Samsung and Aventis. FZ reports personal fees from 89Bio, Abbott, Acceleron, Applied Therapeutics, Bayer, Betagenon, Boehringer, BMS, CVRx, Cambrian, Cardior, Cereno pharmaceutical, Cellprothera, CEVA, Inventiva, KBP, Merck, NovoNordisk, Owkin, Otsuka, Roche Diagnostics, Northsea, USa2, having stock options at G3Pharmaceutical and equities at Cereno, Cardiorenal, Eshmoun Clinical research and being the founder of Cardiovascular Clinical Trialists. MFA received grants from Boehringer-Ingelheim, Gilead, Hanmi, Inventiva, Madrigal, Novo Nordisk; honoraria from Clinical Care Options, Fishawack, Medscape, Terra Firma, CLDF; royalties from Up-to-Date; consults for 89Bio, BMS, Hanmi, Inventiva, Intercept, Madrigal, Merck, Novo Nordisk. SMF received grants from Astellas, Falk Pharma, Genfit, Gilead Sciences, GlympsBio, Janssens Pharmaceutica, Inventiva, Merck Sharp & Dome, Pfizer, Roche; honoraria from Abbvie, Allergan, Bayer, Eisai, Genfit, Gilead Sciences, Janssens Cilag, Intercept, Inventiva, Merck Sharp & Dome, Novo Nordisk, Promethera, Siemens; serves as consultant for Abbvie, Actelion, Aelin Therapeutics, AgomAb, Aligos Therapeutics, Allergan, Alnylam, Astellas, Astra Zeneca, Bayer, Boehringer Ingelheim, Bristoll-Meyers Squibb, CSL Behring, Coherus, Echosens, Dr. Falk Pharma, Eisai, Enyo, Galapagos, Galmed, Genetech, Genfit, Genflow Biosciences, Gilead Sciences, Intercept, Inventiva, Janssens Pharmaceutica, PRO.MED.CS Praha, Julius Clinical, Madrigal, Medimmune, Merck Sharp & Dome, Mursla Bio, NGM Bio, Novartis, Novo Nordisk, Promethera, Roche, Siemens Healthineers.
Figures


References
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

