Molecular Dynamics Study of the Green Solvent Polyethylene Glycol with Water Impurities
- PMID: 38731561
- PMCID: PMC11085543
- DOI: 10.3390/molecules29092070
Molecular Dynamics Study of the Green Solvent Polyethylene Glycol with Water Impurities
Abstract
Polyethylene glycol (PEG) is one of the environmentally benign solvent options for green chemistry. It readily absorbs water when exposed to the atmosphere. The Molecular Dynamics (MD) simulations of PEG200, a commercial mixture of low molecular weight polyethyelene glycol oligomers, as well as di-, tetra-, and hexaethylene glycol are presented to study the effect of added water impurities up to a weight fraction of 0.020, which covers the typical range of water impurities due to water absorption from the atmosphere. Each system was simulated a total of four times using different combinations of two force fields for the water (SPC/E and TIP4P/2005) and two force fields for the PEG and oligomer (OPLS-AA and modified OPLS-AA). The observed trends in the effects of water addition were qualitatively quite robust with respect to these force field combinations and showed that the water does not aggregate but forms hydrogen bonds at most between two water molecules. In general, the added water causes overall either no or very small and nuanced effects in the simulation results. Specifically, the obtained water RDFs are mostly identical regardless of the water content. The added water reduces oligomer hydrogen bonding interactions overall as it competes and forms hydrogen bonds with the oligomers. The loss of intramolecular oligomer hydrogen bonding is in part compensated by oligomers switching from inter- to intramolecular hydrogen bonding. The interplay of the competing hydrogen bonding interactions leads to the presence of shallow extrema with respect to the water weight fraction dependencies for densities, viscosities, and self-diffusion coefficients, in contrast to experimental measurements, which show monotonous dependencies. However, these trends are very small in magnitude and thus confirm the experimentally observed insensitivity of these physical properties to the presence of water impurities.
Keywords: density; ethylene glycol oligomers; hydrogen bonding; polyethylene glycol; radial distribution functions; self diffusion; viscosity; water impurity.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


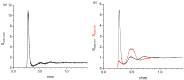
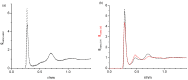

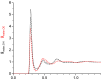


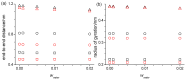
References
-
- Chen J., Spear S.K., Huddleston J.G., Rogers R.D. Polyethylene Glycol and Solutions of Polyethylene Glycol as Green Reaction Media. Green Chem. 2005;7:64–82.
-
- Andrade C.K.Z., Alves L.M. Environmentally Benign Solvents in Organic Synthesis: Current Topics. Curr. Org. Chem. 2005;9:195–218. doi: 10.2174/1385272053369178. - DOI
-
- Hoffmann M.M. Polyethylene glycol as a green chemical solvent. Curr. Opin. Colloid Interface Sci. 2022;57:101537. doi: 10.1016/j.cocis.2021.101537. - DOI
-
- Kardooni R., Kiasat A.R. Polyethylene glycol as a green and biocompatible reaction media for the catalyst free synthesis of organic compounds. Curr. Org. Chem. 2020;24:1275–1314. doi: 10.2174/1385272824999200605161840. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

