Dopamine Release Neuroenergetics in Mouse Striatal Slices
- PMID: 38731799
- PMCID: PMC11083938
- DOI: 10.3390/ijms25094580
Dopamine Release Neuroenergetics in Mouse Striatal Slices
Abstract
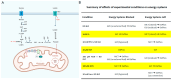
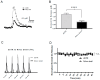
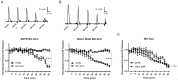
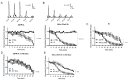
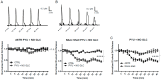
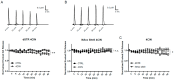
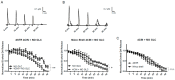
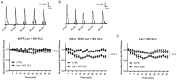
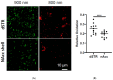
Parkinson's disease (PD) is the second most common neurodegenerative disorder. Dopamine (DA) neurons in the substantia nigra pars compacta, which have axonal projections to the dorsal striatum (dSTR), degenerate in PD. In contrast, DA neurons in the ventral tegmental area, with axonal projections to the ventral striatum, including the nucleus accumbens (NAcc) shell, are largely spared. This study aims to uncover the relative contributions of glycolysis and oxidative phosphorylation (OxPhos) to DA release in the striatum. We measured evoked DA release in mouse striatal brain slices using fast-scan cyclic voltammetry applied every two minutes. Blocking OxPhos resulted in a greater reduction in evoked DA release in the dSTR when compared to the NAcc shell, while blocking glycolysis caused a more significant decrease in evoked DA release in the NAcc shell than in the dSTR. Furthermore, when glycolysis was bypassed in favor of direct OxPhos, evoked DA release in the NAcc shell decreased by approximately 50% over 40 min, whereas evoked DA release in the dSTR was largely unaffected. These results demonstrate that the dSTR relies primarily on OxPhos for energy production to maintain evoked DA release, whereas the NAcc shell depends more on glycolysis. Consistently, two-photon imaging revealed higher oxidation levels of DA terminals in the dSTR than in the NAcc shell. Together, these findings partly explain the selective vulnerability of DA terminals in the dSTR to degeneration in PD.
Keywords: Parkinson’s disease; dopamine release; fast-scan cyclic voltammetry; glycolysis; mitochondria respiration; oxidative phosphorylation.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

