A novel microporous biomaterial vaccine platform for long-lasting antibody mediated immunity against viral infection
- PMID: 38734312
- PMCID: PMC11665867
- DOI: 10.1016/j.jconrel.2024.05.008
A novel microporous biomaterial vaccine platform for long-lasting antibody mediated immunity against viral infection
Abstract
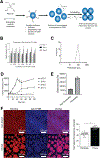
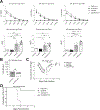
Current antigen delivery platforms, such as alum and nanoparticles, are not readily tunable, thus may not generate optimal adaptive immune responses. We created an antigen delivery platform by loading lyophilized Microporous Annealed Particle (MAP) with aqueous solution containing target antigens. Upon administration of antigen loaded MAP (VaxMAP), the biomaterial reconstitution forms an instant antigen-loaded porous scaffold area with a sustained release profile to maximize humoral immunity. VaxMAP induced CD4+ T follicular helper (Tfh) cells and germinal center (GC) B cell responses in the lymph nodes similar to Alum. VaxMAP loaded with SARS-CoV-2 spike protein improved the magnitude, neutralization, and duration of anti-receptor binding domain antibodies compared to Alum vaccinated mice. A single injection of Influenza specific HA1-loaded-VaxMAP enhanced neutralizing antibodies and elicited greater protection against influenza virus challenge than HA1-loaded-Alum. Thus, VaxMAP is a platform that can be used to promote adaptive immune cell responses to generate more robust neutralizing antibodies, and better protection upon pathogen challenge.
Keywords: Antigen delivery platform; Biomaterial; Sustained release; Vaccine platform.
Copyright © 2024 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest M.N, D.A, W.M.W., S.D. and P.O·S. are shareholders in Tempo Therapeutics, which aims to commercialize MAP technology. P.O.S. and W.M.W. are co-founders of Tempo Therapeutics, and P.O.S. has received a research contract from Tempo Therapeutics unrelated to the work in this manuscript. The authors have no additional financial interests. Correspondence and requests for materials should be addressed to J.S.W. (jason.weinstein@rutgers.edu).
Figures
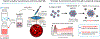



Update of
-
A novel microporous biomaterial vaccine platform for long-lasting antibody mediated immunity against viral infection.bioRxiv [Preprint]. 2024 Jan 31:2024.01.30.578038. doi: 10.1101/2024.01.30.578038. bioRxiv. 2024. Update in: J Control Release. 2024 Jun;370:570-582. doi: 10.1016/j.jconrel.2024.05.008. PMID: 38352398 Free PMC article. Updated. Preprint.
References
-
- Slifka MK, Antia R, Whitmire JK, Ahmed R, Humoral immunity due to long-lived plasma cells, Immunity 8 (1998) 363–372. - PubMed
-
- Cirelli KM, Carnathan DG, Nogal B, Martin JT, Rodriguez OL, Upadhyay AA, Enemuo CA, Gebru EH, Choe Y, Viviano F, Nakao C, Pauthner MG, Reiss S, Cottrell CA, Smith ML, Bastidas R, Gibson W, Wolabaugh AN, Melo MB, Cossette B, Kumar V, Patel NB, Tokatlian T, Menis S, Kulp DW, Burton DR, Murrell B, Schief WR, Bosinger SE, Ward AB, Watson CT, Silvestri G, Irvine DJ, Crotty S, Slow delivery immunization enhances HIV neutralizing antibody and germinal center responses via modulation of Immunodominance, Cell 177 (2019) 1153–1171.e28. - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

