BCKDK modification enhances the anticancer efficacy of CAR-T cells by reprogramming branched chain amino acid metabolism
- PMID: 38734897
- PMCID: PMC11403223
- DOI: 10.1016/j.ymthe.2024.05.017
BCKDK modification enhances the anticancer efficacy of CAR-T cells by reprogramming branched chain amino acid metabolism
Abstract
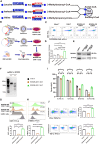
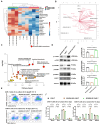
Altered branched chain amino acids (BCAAs), including leucine, isoleucine, and valine, are frequently observed in patients with advanced cancer. We evaluated the efficacy of chimeric antigen receptor (CAR) T cell-mediated cancer cell lysis potential in the immune microenvironment of BCAA supplementation and deletion. BCAA supplementation increased cancer cell killing percentage, while accelerating BCAA catabolism and decreasing BCAA transporter decreased cancer cell lysis efficacy. We thus designed BCKDK engineering CAR T cells for the reprogramming of BCAA metabolism in the tumor microenvironment based on the genotype and phenotype modification. BCKDK overexpression (OE) in CAR-T cells significantly improved cancer cell lysis, while BCKDK knockout (KO) resulted in inferior lysis potential. In an in vivo experiment, BCKDK-OE CAR-T cell treatment significantly prolonged the survival of mice bearing NALM6-GL cancer cells, with the differentiation of central memory cells and an increasing proportion of CAR-T cells in the peripheral circulation. BCKDK-KO CAR-T cell treatment resulted in shorter survival and a decreasing percentage of CAR-T cells in the peripheral circulation. In conclusion, BCKDK-engineered CAR-T cells exert a distinct phenotype for superior anticancer efficiency.
Keywords: BCKDK; branched chain amino acids; cancer metabolism; cancer microenvironment; chimeric antigen receptor; immunotherapy.
Copyright © 2024 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





References
-
- Mullard A. FDA approves fourth CAR-T cell therapy. Nat. Rev. Drug Discov. 2021;20:166. - PubMed
-
- Lin X., Lee S., Sharma P., George B., Scott J. Summary of US Food and Drug Administration Chimeric Antigen Receptor (CAR) T-Cell Biologics License Application Approvals From a Statistical Perspective. J. Clin. Oncol. 2022;40:3501–3509. - PubMed
-
- Granit V., Benatar M., Kurtoglu M., Miljković M.D., Chahin N., Sahagian G., Feinberg M.H., Slansky A., Vu T., Jewell C.M., et al. Safety and clinical activity of autologous RNA chimeric antigen receptor T-cell therapy in myasthenia gravis (MG-001): a prospective, multicentre, open-label, non-randomised phase 1b/2a study. Lancet Neurol. 2023;22:578–590. - PMC - PubMed
-
- Keshavarz A., Salehi A., Khosravi S., Shariati Y., Nasrabadi N., Kahrizi M.S., Maghsoodi S., Mardi A., Azizi R., Jamali S., Fotovat F. Recent findings on chimeric antigen receptor (CAR)-engineered immune cell therapy in solid tumors and hematological malignancies. Stem Cell Res. Ther. 2022;13:482. - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

