Efficacy of neoadjuvant chemo-immunotherapy in non-small cell lung cancer: a real-world, multicenter, retrospective study
- PMID: 38736498
- PMCID: PMC11082708
- DOI: 10.21037/tlcr-24-17
Efficacy of neoadjuvant chemo-immunotherapy in non-small cell lung cancer: a real-world, multicenter, retrospective study
Abstract
Background: Resectable non-small cell lung cancer (NSCLC) patients have a high risk of recurrence. Multiple randomized controlled trials (RCTs) have shown that neoadjuvant chemo-immunotherapy brings new hope for these patients. The study aims to evaluate the safety, surgery-related outcomes and oncological outcomes for neoadjuvant chemo-immunotherapy in real-world setting with a large sample size and long-term follow-up.
Methods: Patients with clinical stage IB-IIIB NSCLC who received neoadjuvant chemo-immunotherapy at two Chinese institutions were included in this retrospective cohort study. Surgical and oncological outcomes of the enrolled NSCLC patients were collected and analyzed.
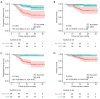
Results: There were 158 patients identified, of which 124 (78.5%) were at stage IIIA-IIIB and the remaining 34 (21.5%) were at stage IB-IIB. Forty-one patients (25.9%) received two cycles of neoadjuvant treatment, 80 (50.6%) had three cycles, and 37 (23.4%) had four cycles. Twenty-four patients (15.2%) experienced grade 3 or worse immune-related adverse events. The median interval time between the last neoadjuvant therapy and surgery was 37 [interquartile range (IQR), 31-43] days. Fifty-eight out of 96 (60.4%) central NSCLC patients who were expected to undergo complex surgery had the scope or the difficulty of operation reduced. Ninety-five (60.1%) patients achieved major pathologic response (MPR), including 62 (39.2%) patients with pathologic complete response (pCR). Multivariate regression analysis showed that no clinical factor other than programmed death-ligand 1 (PD-L1) expression was predictive of the pathological response. The median follow-up time from diagnosis was 27.1 months. MPR and pCR were significantly associated with improved progression-free survival (PFS) and overall survival (OS). Neither stage nor PD-L1 expression was significantly associated with long-term survival.
Conclusions: The neoadjuvant chemo-immunotherapy is a feasible strategy for NSCLC with a favorable rate of pCR/MPR, modified resection and 2-year survival. No clinical factor other than PD-L1 expression was predictive of the pathological response. pCR/MPR may be effective surrogate endpoint for survival in NSCLC patients who received neoadjuvant chemo-immunotherapy.
Keywords: Non-small cell lung cancer (NSCLC); chemo-immunotherapy; neoadjuvant therapy; programmed death-1/programmed death-ligand 1 (PD-1/PD-L1); surgical extent.
2024 Translational Lung Cancer Research. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-24-17/coif). The authors have no conflicts of interest to declare.
Figures

References
LinkOut - more resources
Full Text Sources
Research Materials
