Longitudinal assessment of real-world patient adherence: a 12-month electronic patient-reported outcomes follow-up of women with early breast cancer undergoing treatment
- PMID: 38740611
- PMCID: PMC11090970
- DOI: 10.1007/s00520-024-08547-7
Longitudinal assessment of real-world patient adherence: a 12-month electronic patient-reported outcomes follow-up of women with early breast cancer undergoing treatment
Abstract
Background: Electronic patient-reported outcomes (ePROs) assess patients' health status and quality of life, improving patient care and treatment effects, yet little is known about their use and adherence in routine patient care.
Aims: We evaluated the adherence of invasive breast cancer and ductal carcinoma in situ (DCIS) patients to ePROs follow-up and whether specific patient characteristics are related to longitudinal non-adherence.
Methods: Since November 2016, the Breast Center at Charité - Universitätsmedizin Berlin has implemented an ongoing prospective PRO routine program, requiring patients to complete ePROs assessments and consent to email-based follow-up in the first 12 months after therapy starts. Frequencies and summary statistics are presented. Multiple logistic regression models were performed to determine an association between patient characteristics and non-adherence.
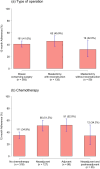
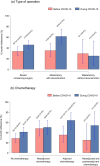
Results: Out of 578 patients, 239 patients (41.3%, 95%CI: 37.3-45.5%) completed baseline assessment and all five ePROs follow-up during the first 12 months after therapy. On average, above 70% of those patients responded to the ePROs follow-up assessment. Adherence to the ePROs follow-up was higher during the COVID-19 pandemic than in the time periods before (47.4% (111/234) vs. 33.6% (71/211)). Factors associated with longitudinal non-adherence were younger age, a higher number of comorbidities, no chemotherapy, and a low physical functioning score in the EORTC QLQ-C30 at baseline.
Conclusions: The study reveals moderate adherence to 12-month ePROs follow-up assessments in invasive early breast cancer and DCIS patients, with response rates ranging from 60 to 80%. Emphasizing the benefits for young patients and those with high disease burdens might further increase adherence.
Keywords: Compliance; Early Breast cancer; Oncology; Return rate; Web-based questionnaire.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
The authors declare no competing interests.
Figures

References
-
- Singh K, Landman AB (2017) Chapter 13 - mobile health. In: Sheikh A, Cresswell KM, Wright A, et al (eds). Key advances in clinical informatics. Academic Press, pp 183-196. 10.1016/B978-0-12-809523-2.00013-3
-
- Mody GN, Stover AM, Wang M, King-Kallimanis BL, Jansen J, Henson S, et al. Electronic patient-reported outcomes monitoring during lung cancer chemotherapy: A nested cohort within the PRO-TECT pragmatic trial (AFT-39) Lung Cancer. 2021;162:1–8. doi: 10.1016/j.lungcan.2021.09.020. - DOI - PMC - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

