Recent advancements in cartilage tissue engineering innovation and translation
- PMID: 38740860
- PMCID: PMC11524031
- DOI: 10.1038/s41584-024-01118-4
Recent advancements in cartilage tissue engineering innovation and translation
Abstract
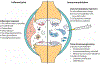
Articular cartilage was expected to be one of the first successfully engineered tissues, but today, cartilage repair products are few and they exhibit considerable limitations. For example, of the cell-based products that are available globally, only one is marketed for non-knee indications, none are indicated for severe osteoarthritis or rheumatoid arthritis, and only one is approved for marketing in the USA. However, advances in cartilage tissue engineering might now finally lead to the development of new cartilage repair products. To understand the potential in this field, it helps to consider the current landscape of tissue-engineered products for articular cartilage repair and particularly cell-based therapies. Advances relating to cell sources, bioactive stimuli and scaffold or scaffold-free approaches should now contribute to progress in therapeutic development. Engineering for an inflammatory environment is required because of the need for implants to withstand immune challenge within joints affected by osteoarthritis or rheumatoid arthritis. Bringing additional cartilage repair products to the market will require an understanding of the translational vector for their commercialization. Advances thus far can facilitate the future translation of engineered cartilage products to benefit the millions of patients who suffer from cartilage injuries and arthritides.
© 2024. Springer Nature Limited.
Conflict of interest statement
Competing interests
Drs. Athanasiou and Hu have an equity interest in Cartilage, Inc. Their relationship with Cartilage, Inc. has been reviewed and approved by the University of California, Irvine in accordance with its conflict-of-interest policies.
Figures


References
-
- Langer R & Vacanti JP Tissue Engineering. Science (1979) 260, 920–926 (1993). - PubMed
-
- Matthews LS, Sonstegard DA & Henke JA Load Bearing Characteristics of the Patello-Femoral Joint. Acta Orthop Scand 48, 511–516 (1977). - PubMed
-
- Kellathur SN & Lou H-X Cell and tissue therapy regulation: Worldwide status and harmonization. Biologicals 40, 222–224 (2012). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

