Actionability and familial uptake following opportunistic genomic screening in a pediatric cancer cohort
- PMID: 38740897
- PMCID: PMC11220050
- DOI: 10.1038/s41431-024-01618-7
Actionability and familial uptake following opportunistic genomic screening in a pediatric cancer cohort
Abstract
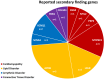
The care for patients with serious conditions is increasingly guided by genomic medicine, and genomic medicine may equally transform care for healthy individual if genomic population screening is implemented. This study examines the medical impact of opportunistic genomic screening (OGS) in a cohort of patients undergoing comprehensive genomic germline DNA testing for childhood cancer, including the impact on their relatives. Medical actionability and uptake after cascade testing in the period following disclosure of OGS results was quantified. A secondary finding was reported to 19/595 (3.2%) probands primarily in genes related to cardiovascular and lipid disorders. After a mean follow up time of 1.6 years (Interquartile range (IQR): 0.57-1.92 yrs.) only 12 (63%) of these variants were found to be medically actionable. Clinical follow up or treatment was planned in 16 relatives, and as in the probands, the prescribed treatment was primarily betablockers or cholesterol lowering therapy. No invasive procedures or implantation of medical devices were performed in probands or relatives, and no reproductive counseling was requested. After an average of 1.6 years of follow-up 2.25 relatives per family with an actionable finding had been tested. This real-world experience of OGS grants new insight into the practical implementation effects and derived health care demands of genotype-first screening. The resulting health care effect and impact on demand for genetic counseling and workup in relatives extends beyond the effect in the probands.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Familial communication and cascade testing following elective genomic testing.J Genet Couns. 2025 Feb;34(1):e1907. doi: 10.1002/jgc4.1907. Epub 2024 May 16. J Genet Couns. 2025. PMID: 38757439
-
The Impact of Proband Indication for Genetic Testing on the Uptake of Cascade Testing Among Relatives.Front Genet. 2022 Jun 16;13:867226. doi: 10.3389/fgene.2022.867226. eCollection 2022. Front Genet. 2022. PMID: 35783293 Free PMC article.
-
Long-Term Follow-Up Study on the Uptake of Genetic Counseling and Predictive DNA Testing in Inherited Cardiac Conditions.Circ Genom Precis Med. 2020 Oct;13(5):524-530. doi: 10.1161/CIRCGEN.119.002803. Epub 2020 Aug 14. Circ Genom Precis Med. 2020. PMID: 33079600 Free PMC article.
-
The uptake and utility of genetic testing and genetic counseling for hypertrophic cardiomyopathy-A systematic review and meta-analysis.J Genet Couns. 2022 Dec;31(6):1290-1305. doi: 10.1002/jgc4.1604. Epub 2022 Jul 7. J Genet Couns. 2022. PMID: 35799446
-
Genetic Testing for Familial Hypercholesterolemia: Health Technology Assessment.Ont Health Technol Assess Ser. 2022 Aug 23;22(3):1-155. eCollection 2022. Ont Health Technol Assess Ser. 2022. PMID: 36158868 Free PMC article.
Cited by
-
Hitting the heights with CiteScore.Eur J Hum Genet. 2024 Jul;32(7):743-744. doi: 10.1038/s41431-024-01651-6. Eur J Hum Genet. 2024. PMID: 38956180 Free PMC article. No abstract available.
-
Parental perspectives on the use of tumor molecular profiling and germline genetic testing during their children's cancer treatment.Fam Cancer. 2025 Aug 11;24(3):63. doi: 10.1007/s10689-025-00488-2. Fam Cancer. 2025. PMID: 40788353
References
-
- Kalia SS, Adelman K, Bale SJ, Chung WK, Eng C, Evans JP, et al. Recommendations for reporting of secondary findings in clinical exome and genome sequencing, 2016 update (ACMG SF v2.0): A policy statement of the American College of Medical Genetics and Genomics. Genet Med. 2017;19:249–55. doi: 10.1038/gim.2016.190. - DOI - PubMed
-
- Miller DT, Lee K, Abul-Husn NS, Amendola LM, Brothers K, Chung WK, et al. ACMG SF v3.1 list for reporting of secondary findings in clinical exome and genome sequencing: A policy statement of the American College of Medical Genetics and Genomics (ACMG) Genet Med. 2022;24:1407–14. doi: 10.1016/j.gim.2022.04.006. - DOI - PubMed
-
- Miller DT, Lee K, Chung WK, Gordon AS, Herman GE, Klein TE, et al. ACMG SF v3.0 list for reporting of secondary findings in clinical exome and genome sequencing: a policy statement of the American College of Medical Genetics and Genomics (ACMG) Genet Med. 2021;23:1381–90. doi: 10.1038/s41436-021-01172-3. - DOI - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

