Bax Inhibitor-1 preserves pancreatic β-cell proteostasis by limiting proinsulin misfolding and programmed cell death
- PMID: 38744890
- PMCID: PMC11094198
- DOI: 10.1038/s41419-024-06701-x
Bax Inhibitor-1 preserves pancreatic β-cell proteostasis by limiting proinsulin misfolding and programmed cell death
Abstract
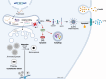
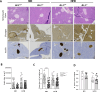
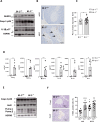
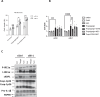
The prevalence of diabetes steadily increases worldwide mirroring the prevalence of obesity. Endoplasmic reticulum (ER) stress is activated in diabetes and contributes to β-cell dysfunction and apoptosis through the activation of a terminal unfolded protein response (UPR). Our results uncover a new role for Bax Inhibitor-One (BI-1), a negative regulator of inositol-requiring enzyme 1 (IRE1α) in preserving β-cell health against terminal UPR-induced apoptosis and pyroptosis in the context of supraphysiological loads of insulin production. BI-1-deficient mice experience a decline in endocrine pancreatic function in physiological and pathophysiological conditions, namely obesity induced by high-fat diet (HFD). We observed early-onset diabetes characterized by hyperglycemia, reduced serum insulin levels, β-cell loss, increased pancreatic lipases and pro-inflammatory cytokines, and the progression of metabolic dysfunction. Pancreatic section analysis revealed that BI-1 deletion overburdens unfolded proinsulin in the ER of β-cells, confirmed by ultrastructural signs of ER stress with overwhelmed IRE1α endoribonuclease (RNase) activity in freshly isolated islets. ER stress led to β-cell dysfunction and islet loss, due to an increase in immature proinsulin granules and defects in insulin crystallization with the presence of Rod-like granules. These results correlated with the induction of autophagy, ER phagy, and crinophagy quality control mechanisms, likely to alleviate the atypical accumulation of misfolded proinsulin in the ER. In fine, BI-1 in β-cells limited IRE1α RNase activity from triggering programmed β-cell death through apoptosis and pyroptosis (caspase-1, IL-1β) via NLRP3 inflammasome activation and metabolic dysfunction. Pharmaceutical IRE1α inhibition with STF-083010 reversed β-cell failure and normalized the metabolic phenotype. These results uncover a new protective role for BI-1 in pancreatic β-cell physiology as a stress integrator to modulate the UPR triggered by accumulating unfolded proinsulin in the ER, as well as autophagy and programmed cell death, with consequences on β-cell function and insulin secretion. In pancreatic β-cells, BI-1-/- deficiency perturbs proteostasis with proinsulin misfolding, ER stress, terminal UPR with overwhelmed IRE1α/XBP1s/CHOP activation, inflammation, β-cell programmed cell death, and diabetes.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Fernandes-da-Silva A, Miranda CS, Santana-Oliveira DA, Oliveira-Cordeiro B, Rangel-Azevedo C, Silva-Veiga FM, et al. Endoplasmic reticulum stress as the basis of obesity and metabolic diseases: focus on adipose tissue, liver, and pancreas. Eur J Nutr. 2021;60:2949–60. doi: 10.1007/s00394-021-02542-y. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

