Physiologically based absorption modeling to predict the bioequivalence of two apixaban formulations
- PMID: 38747478
- PMCID: PMC11095081
- DOI: 10.1111/cts.13819
Physiologically based absorption modeling to predict the bioequivalence of two apixaban formulations
Abstract
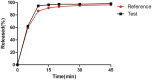
The equivalence of absorption rates and extents between generic drugs and their reference formulations is crucial for ensuring therapeutic comparability. Bioequivalence (BE) studies are widely utilized and play a pivotal role in substantiating the approval and promotional efforts for generic drugs. Virtual BE simulation is a valuable tool for mitigating risks and guiding clinical BE studies, thereby minimizing redundant in vivo BE assessments. Herein, we successfully developed a physiologically based absorption model for virtual BE simulations, which precisely predicts the BE of the apixaban test and reference formulations. The modeling results confirm that the test and reference formulations were bioequivalent under both fasted and fed conditions, consistent with clinical studies. This highlights the efficacy of physiologically based absorption modeling as a powerful tool for formulation screening and can be adopted as a methodological and risk assessment strategy to detect potential clinical BE risks.
© 2024 The Author(s). Clinical and Translational Science published by Wiley Periodicals LLC on behalf of American Society for Clinical Pharmacology and Therapeutics.
Conflict of interest statement
The authors declared no competing interests for this work.
Figures
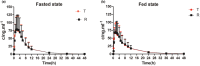
Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Oral direct thrombin inhibitors or oral factor Xa inhibitors for the treatment of deep vein thrombosis.Cochrane Database Syst Rev. 2015 Jun 30;(6):CD010956. doi: 10.1002/14651858.CD010956.pub2. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2023 Apr 14;4:CD010956. doi: 10.1002/14651858.CD010956.pub3. PMID: 26123214 Updated.
-
Virtual twin approach using physiologically based pharmacokinetic modelling in hospitalized patients treated with apixaban or rivaroxaban.Br J Clin Pharmacol. 2025 Jul;91(7):2057-2069. doi: 10.1002/bcp.70032. Epub 2025 Mar 4. Br J Clin Pharmacol. 2025. PMID: 40035306
-
Bioequivalence of generic and branded ibrutinib capsules in healthy Chinese volunteers under fasting and fed conditions: a randomized, four-period, fully replicated, crossover study.Expert Opin Drug Metab Toxicol. 2025 Jul;21(7):875-883. doi: 10.1080/17425255.2025.2496459. Epub 2025 Apr 28. Expert Opin Drug Metab Toxicol. 2025. PMID: 40264436 Clinical Trial.
-
Non-vitamin K antagonist oral anticoagulants (NOACs) after transcatheter aortic valve replacement (TAVR): a network meta-analysis.Cochrane Database Syst Rev. 2025 Feb 24;2(2):CD013745. doi: 10.1002/14651858.CD013745.pub2. Cochrane Database Syst Rev. 2025. PMID: 39991882
References
-
- U.S. Department of Health and Human Services , Food and Drug Administration (FDA) , Center for Evaluation and Research (CDER) . Bioequivalence Studies with Pharmacokinetic Endpoints for Drugs Submitted Under an ANDA. Guidance for Industry; Draft guidance. https://www.fda.gov/media/87219/download. Accessed August, 2021.
-
- Miao L, Mousa YM, Zhao L, Raines K, Seo P, Wu F. Using a physiologically based pharmacokinetic absorption model to establish dissolution bioequivalence safe space for oseltamivir in adult and pediatric populations. AAPS J. 2020;22:107. - PubMed
-
- Wu F, Cristofoletti R, Zhao L, Rostami‐Hodjegan A. Scientific considerations to move towards biowaiver for biopharmaceutical classification system class III drugs: how modeling and simulation can help. Biopharm Drug Dispos. 2021;42:118‐127. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

