The effect of current antithrombotic therapy on mortality in nursing home residents with COVID-19: a multicentre retrospective cohort study
- PMID: 38748450
- PMCID: PMC11095411
- DOI: 10.1093/ageing/afae094
The effect of current antithrombotic therapy on mortality in nursing home residents with COVID-19: a multicentre retrospective cohort study
Abstract
Background: The first wave of COVID led to an alarmingly high mortality rate among nursing home residents (NHRs). In hospitalised patients, the use of anticoagulants may be associated with a favourable prognosis. However, it is unknown whether the use of antithrombotic medication also protected NHRs from COVID-19-related mortality.
Objectives: To investigate the effect of current antithrombotic therapy in NHRs with COVID-19 on 30-day all-cause mortality during the first COVID-19 wave.
Methods: We performed a retrospective cohort study linking electronic health records and pharmacy data in NHRs with COVID-19. A propensity score was used to match NHRs with current use of therapeutic dose anticoagulants to NHRs not using anticoagulant medication. The primary outcome was 30-day all-cause mortality, which was evaluated using a logistic regression model. In a secondary analysis, multivariable logistic regression was performed in the complete study group to compare NHRs with current use of therapeutic dose anticoagulants and those with current use of antiplatelet therapy to those without such medication.
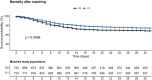
Results: We included 3521 NHRs with COVID-19 based on a positive RT-PCR for SARS-CoV-2 or with a well-defined clinical suspicion of COVID-19. In the matched propensity score analysis, NHRs with current use of therapeutic dose anticoagulants had a significantly lower all-cause mortality (OR = 0.73; 95% CI: 0.58-0.92) compared to NHRs who did not use therapeutic anticoagulants. In the secondary analysis, current use of therapeutic dose anticoagulants (OR: 0.62; 95% CI: 0.48-0.82) and current use of antiplatelet therapy (OR 0.80; 95% CI: 0.64-0.99) were both associated with decreased mortality.
Conclusions: During the first COVID-19 wave, therapeutic anticoagulation and antiplatelet use were associated with a reduced risk of all-cause mortality in NHRs. Whether these potentially protective effects are maintained in vaccinated patients or patients with other COVID-19 variants, remains unknown.
Keywords: COVID-19; anticoagulants; antiplatelets; antithrombotic medication; mortality; nursing home residents; older people.
© The Author(s) 2024. Published by Oxford University Press on behalf of the British Geriatrics Society.
Conflict of interest statement
Firdaouss Boutkourt, Thijs van Haaps, Reneé Brüggemann, Soerajja Bhoelan, Bart Spaetgens, Nick van Es, Tineke Roest, Karlijn J. Joling and Jacqueline Hugtenburg have no relevant financial or non-financial interests to disclose. Dr M.J.H.A. Kruip received speakers fee from Sobi, BMS and Roche all paid to the institution (Erasmus MC) and an unrestricted grant by Sobi, payment to institution (Erasmus MC). Dr Meijer reports speaker fees from Alexion, Bayer and CSL Behring, participation in trial steering committees for Bayer and Astra Zeneca, consulting fees from Uniqure, participation in data monitoring and endpoint adjudication committee for Octapharma. Dr Hugo ten Cate received consultancy fees from Alveron, Novostia, Astra Zeneca and Galapagos and is a shareholder at Coagulation Profile; all revenues deposited at the CARIM institute for research.
Figures


References
-
- Investigators I, Sadeghipour P, Talasaz AHet al. . Effect of intermediate-dose vs standard-dose prophylactic anticoagulation on thrombotic events, extracorporeal membrane oxygenation treatment, or mortality among patients with COVID-19 admitted to the intensive care unit: the INSPIRATION randomized clinical trial. JAMA 2021; 325: 1620–30. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

