Contribution of genetic polymorphism in ABCB1 to individual variations of imatinib plasma levels in patients with gastrointestinal stromal tumor
- PMID: 38756645
- PMCID: PMC11094496
- DOI: 10.21037/jgo-24-188
Contribution of genetic polymorphism in ABCB1 to individual variations of imatinib plasma levels in patients with gastrointestinal stromal tumor
Abstract
Background: Imatinib mesylate (IM) is a first-line treatment option for the majority of patients diagnosed with gastrointestinal stromal tumors (GISTs). Although the clinical benefit is high, interindividual response is variable. This study thus aimed to assess how genetic polymorphisms can affect the blood levels of IM and treatment outcomes in patients with GIST.
Methods: A total of 31 single-nucleotide polymorphisms (SNPs) in selected cytochrome P450 (P450), ATP-binding cassette transporter (ABC), solute carrier family (SLC), interleukin-4 receptor (IL4R), and vascular endothelial growth factor (VEGF) genes were genotyped using an SNP mass array platform. A total of 192 consecutive patients with GIST who received 400 mg of IM daily were enrolled into the study, with 1,485 blood samples being analyzed. According to genotypes, IM trough concentrations were tested and compared. Progression-free survival (PFS) and overall survival (OS) were also assessed.
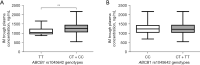
Results: With a mean follow-up of 75.99 months, trough concentrations of imatinib were examined at average time points of 7.73 for each patient. Polymorphism in ABCB1 rs1045642 was found to be associated with steady-state IM trough plasma levels (P=0.008). Patients with the C genotype (CT + CC) of rs1045642 exhibited higher IM trough concentrations (1,271.09±306.69 ng/mL) compared to those with the TT genotype (1,106.60±206.05 ng/mL). No statistically significant differences in IM plasma concentration were observed for the other SNPs tested. None of the tested SNPs displayed a significant association with patients' survival in this study.
Conclusions: This is the largest cohort study evaluating the associations of SNP and imatinib blood trough levels. The ABCB1 rs1045642 genetic polymorphism may exert an effect on the pharmacokinetics of imatinib. The presence of the C allele in ABCB1 rs1045642 is predictive of a higher plasma concentration of IM.
Keywords: ABCB1; Imatinib; gastrointestinal stromal tumor (GIST); single-nucleotide polymorphism (SNP).
2024 Journal of Gastrointestinal Oncology. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jgo.amegroups.com/article/view/10.21037/jgo-24-188/coif). The authors have no conflicts of interest to declare.
Figures
Similar articles
-
Genetic Polymorphisms Contribute to the Individual Variations of Imatinib Mesylate Plasma Levels and Adverse Reactions in Chinese GIST Patients.Int J Mol Sci. 2017 Mar 13;18(3):603. doi: 10.3390/ijms18030603. Int J Mol Sci. 2017. PMID: 28335376 Free PMC article.
-
Impact of ABCG2 and ABCB1 Polymorphisms on Imatinib Plasmatic Exposure: An Original Work and Meta-Analysis.Int J Mol Sci. 2023 Feb 7;24(4):3303. doi: 10.3390/ijms24043303. Int J Mol Sci. 2023. PMID: 36834713 Free PMC article.
-
Association of ABCG2 polymorphism with clinical efficacy of imatinib in patients with gastrointestinal stromal tumor.Cancer Chemother Pharmacol. 2015 Jan;75(1):173-82. doi: 10.1007/s00280-014-2630-6. Epub 2014 Nov 23. Cancer Chemother Pharmacol. 2015. PMID: 25417047
-
Influence of genetic polymorphisms on imatinib concentration and therapeutic response in patients with chronic-phase chronic myeloid leukemia.Int Immunopharmacol. 2024 May 30;133:112090. doi: 10.1016/j.intimp.2024.112090. Epub 2024 Apr 18. Int Immunopharmacol. 2024. PMID: 38640718
-
Effect of CYP3A and ABCB1 single nucleotide polymorphisms on the pharmacokinetics and pharmacodynamics of calcineurin inhibitors: Part I.Clin Pharmacokinet. 2010 Mar;49(3):141-75. doi: 10.2165/11317350-000000000-00000. Clin Pharmacokinet. 2010. PMID: 20170205 Review.
Cited by
-
ETV1 genetic polymorphisms as a candidate prognosis biomarker of Gastrointestinal stromal tumor.Cancer Chemother Pharmacol. 2025 Jul 2;95(1):68. doi: 10.1007/s00280-025-04789-2. Cancer Chemother Pharmacol. 2025. PMID: 40601091
References
LinkOut - more resources
Full Text Sources
