Exogenous erythropoietin increases hematological status, fat oxidation, and aerobic performance in males following prolonged strenuous training
- PMID: 38757249
- PMCID: PMC11099744
- DOI: 10.14814/phy2.16038
Exogenous erythropoietin increases hematological status, fat oxidation, and aerobic performance in males following prolonged strenuous training
Abstract
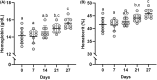
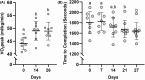
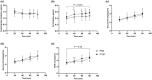
This study investigated the effects of EPO on hemoglobin (Hgb) and hematocrit (Hct), time trial (TT) performance, substrate oxidation, and skeletal muscle phenotype throughout 28 days of strenuous exercise. Eight males completed this longitudinal controlled exercise and feeding study using EPO (50 IU/kg body mass) 3×/week for 28 days. Hgb, Hct, and TT performance were assessed PRE and on Days 7, 14, 21, and 27 of EPO. Rested/fasted muscle obtained PRE and POST EPO were analyzed for gene expression, protein signaling, fiber type, and capillarization. Substrate oxidation and glucose turnover were assessed during 90-min of treadmill load carriage (LC; 30% body mass; 55 ± 5% V̇O2peak) exercise using indirect calorimetry, and 6-6-[2H2]-glucose PRE and POST. Hgb and Hct increased, and TT performance improved on Days 21 and 27 compared to PRE (p < 0.05). Energy expenditure, fat oxidation, and metabolic clearance rate during LC increased (p < 0.05) from PRE to POST. Myofiber type, protein markers of mitochondrial biogenesis, and capillarization were unchanged PRE to POST. Transcriptional regulation of mitochondrial activity and fat metabolism increased from PRE to POST (p < 0.05). These data indicate EPO administration during 28 days of strenuous exercise can enhance aerobic performance through improved oxygen carrying capacity, whole-body and skeletal muscle fat metabolism.
Keywords: V̇O2peak; hematocrit; hemoglobin; mitochondria; substrate oxidation.
© 2024 The Authors. Physiological Reports published by Wiley Periodicals LLC on behalf of The Physiological Society and the American Physiological Society.
Conflict of interest statement
The authors have nothing to disclose.
Figures


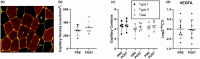

Similar articles
-
Strenuous training combined with erythropoietin induces red cell volume expansion-mediated hypervolemia and alters systemic and skeletal muscle iron homeostasis.Am J Physiol Regul Integr Comp Physiol. 2024 Nov 1;327(5):R473-R478. doi: 10.1152/ajpregu.00164.2024. Epub 2024 Sep 6. Am J Physiol Regul Integr Comp Physiol. 2024. PMID: 39241004
-
Erythropoietin enhances whole body lipid oxidation during prolonged exercise in humans.J Physiol Biochem. 2015 Mar;71(1):9-16. doi: 10.1007/s13105-014-0374-8. Epub 2015 Jan 9. J Physiol Biochem. 2015. PMID: 25567744
-
Effects of an 8-weeks erythropoietin treatment on mitochondrial and whole body fat oxidation capacity during exercise in healthy males.J Sports Sci. 2015;33(6):570-8. doi: 10.1080/02640414.2014.951872. Epub 2014 Sep 26. J Sports Sci. 2015. PMID: 25259652
-
Regulation of skeletal muscle fat oxidation during exercise in humans.Med Sci Sports Exerc. 2002 Sep;34(9):1477-84. doi: 10.1097/00005768-200209000-00013. Med Sci Sports Exerc. 2002. PMID: 12218742 Review.
-
The regulation of carbohydrate and fat metabolism during and after exercise.Front Biosci. 1998 Sep 15;3:D1011-27. doi: 10.2741/a342. Front Biosci. 1998. PMID: 9740552 Review.
Cited by
-
Strenuous training combined with erythropoietin induces red cell volume expansion-mediated hypervolemia and alters systemic and skeletal muscle iron homeostasis.Am J Physiol Regul Integr Comp Physiol. 2024 Nov 1;327(5):R473-R478. doi: 10.1152/ajpregu.00164.2024. Epub 2024 Sep 6. Am J Physiol Regul Integr Comp Physiol. 2024. PMID: 39241004
References
-
- American College of Sports Medicine (2006). Guidelines for exercise testing and prescription (7th ed.). Wolters Kluwer.
-
- Annaheim, S. , Jacob, M. , Krafft, A. , Breymann, C. , Rehm, M. , & Boutellier, U. (2016). RhEPO improves time to exhaustion by non‐hematopoietic factors in humans. European Journal of Applied Physiology, 116, 623–633. - PubMed
-
- Ashby, D. R. , Gale, D. P. , Busbridge, M. , Murphy, K. G. , Duncan, N. D. , Cairns, T. D. , Taube, D. H. , Bloom, S. R. , Tam, F. W. , Chapman, R. , Maxwell, P. H. , & Choi, P. (2010). Erythropoietin administration in humans causes a marked and prolonged reduction in circulating hepcidin. Haematologica, 95, 505–508. - PMC - PubMed
-
- Bellinger, P. M. , Sabapathy, S. , Craven, J. , Arnold, B. , & Minahan, C. (2020). Overreaching attenuates training‐induced improvements in muscle oxidative capacity. Medicine and Science in Sports and Exercise, 52, 77–85. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

